SUPPLEMENTARY INFORMATION
|
|
|
- Rodney Shepherd
- 6 years ago
- Views:
Transcription
1 SUPPLEMENTARY INFRMATIN Acid- labile mpeg- Vinyl Ether- 1,2- Dioleylglycerol Lipids with Tunable ph Sensitivity: Synthesis and Structural Effects on Hydrolysis Rates, DPE Liposome Release Performance and Pharmacokinetics Junhwa Shin, Pochi Shum, Jessica Grey, Shin- ichi Fujiwara, Guarov S. Malhotra, Andres González- Bonet, Seok- Hee Hyun, Elaine Moase, Teresa M. Allen, & David H. Thompson * Table of Contents 1. Details of DFT Computations 2 2. ptimized Structures and Energies for CURVE Synthesis of mpeg- Vinyl Ether Lipids a. Synthesis route for Me- PEG- 1 - oxy- 1 - propene ,2- dioleylglycerol (ST902 & ST905), Figure S1 10 b. Synthesis route for Me- PEG- 1 - oxyethene ,2- dioleylglycerol (ST912 & ST915), Figure S2 10 c. Synthesis route for Me- PEG- 1 - oxy- 2 - propene ,2- dioleylglycerol (ST502 & ST505), Figure S3 11 d. Synthesis route for Me- PEG- 1 - amido- 2 - propene ,2- dioleylglycerol (ST152 & ST155), Figure S Detailed Description of Me- PEG- VE- DG Lipid Syntheses Hydrolysis Studies by TLC, Figure S Hydrolysis Studies by HPLC & 1 H NMR, Figures S Calcein Release Rates for Various mpeg- VE- lipid Loadings in DPE Liposomes a. DPE with ST155 and ST305 Figure S12 23 b. DPE with ST502 and ST505, Figure S References 25 1
2 1. Details of DFT Computations Density functional theory (DFT) calculations were used to determine the proton affinity (PA) values of a series of vinyl ethers using Gaussian The DFT method was employed using the B3LYP hybrid functional. 2,3 Neutral and protonated structures were optimized with 6-31G** basis set. Single point energies of the optimized structures were obtained with the cc- pvtz basis set. 4 The PA is the negative of the standard enthalpy variation ( rh ) for the reaction, R R Y Me + Y + Me 1 2 H + where PA(M) = - rh 298K. Table S1. PA values of a series of methyl vinyl ethers 1. CURVE R Y Neutral (hartree) Protonated (hartree) PA (kcal/mol) 1a Me H b H Me c H Ph d H C()Me e H Me f Ph Me g Me Me h H C()NHMe i H CH 2 C()NHMe
3 2. ptimized Structures and Energies of CURVE 1a (neutral) SCF Done: E(RB+HF- LYP) = A.U. after 7 cycles a (protonated) SCF Done: E(RB+HF- LYP) = A.U. after 7 cycles
4 1b (neutral) SCF Done: E(RB+HF- LYP) = A.U. after 6 cycles b (protonated) SCF Done: E(RB+HF- LYP) = A.U. after 6 cycles
5 1c (neutral) SCF Done: E(RB+HF- LYP) = A.U. after 7 cycles c (protonated) SCF Done: E(RB+HF- LYP) = A.U. after 8 cycles
6 1d (neutral) SCF Done: E(RB+HF- LYP) = A.U. after 8 cycles d (protonated) SCF Done: E(RB+HF- LYP) = A.U. after 8 cycles
7 1e (neutral) SCF Done: E(RB+HF- LYP) = A.U. after 7 cycles e (protonated) SCF Done: E(RB+HF- LYP) = A.U. after 7 cycles
8 1f (neutral) SCF Done: E(RB+HF- LYP) = A.U. after 8 cycles f (protonated) SCF Done: E(RB+HF- LYP) = A.U. after 8 cycles
9 1g (neutral) SCF Done: E(RB+HF- LYP) = A.U. after 7 cycles g (protonated) SCF Done: E(RB+HF- LYP) = A.U. after 7 cycles
10 3. Synthesis of mpeg- Vinyl Ether Lipids 3a. Synthesis route for Me- PEG- 1 - oxyethene ,2- dioleylglycerol (ST912 & 915). C 18 H 35 C C 18 H 18 H C 1) t-buk 18 H Pd(Ac) 2, N N 35 C 18 H C 18 H ) ptsh ethyl vinyl ether H 87% 81% C 18 H 35 C 18 H 35 Me NIS n H I Me n 7 (PEG2000 Conjugate, 44% yield) 8 (PEG5000 Conjugate, 52% yield) t-buk Me n ST912 (75%) ST915 (94%) 3b. Synthesis route for Me- PEG- 1 - oxy- 1 - propene ,2- dioleylglycerol (ST902 & 905). C 18 H 35 C 18 H 35 C 18 H 35 C 18 H 35 t-buk DMS C 18 H 35 93% 1 2 Me NIS n t-buk H C 18 H 35 I Me 3 (PEG2000 Conjugate, 55% yield) 4 (PEG5000 Conjugate, 41% yield) n Me n ST902 (92%) ST905 (69%) 10
11 3c. Synthesis route for Me- PEG- 1 - oxy- 2 - propene ,2- dioleylglycerol (ST502 & 505). C 18 H 35 TBDMS TBDMS C 18 H 35 C 18 H 35 TiCl 4, CH 2 Br 2, C 18 H 35 C 18 H 35 C 18 H 35 (CCl) 2 Zn, TMEDA TBDMS H 65% 42% TBDMS 92% TBAF C 18 H 35 C 18 H 35 PNPCCl, Et 3 N C 18 H 35 C 18 H 35 CPNP 92% H Me n NH 2 Me n H N ST502 (83%) ST505 (71%) 3d. Synthesis route for Me- PEG- 1 - amido- 2 - propene ,2- dioleylglycerol (ST152 & 155). C 18 H 35 C 18 H 35 C 18 H 35 C 18 H 35 N 2 CH 2 N(CH 3 ) 2 I C 18 H 35 C 18 H 35 C(C 2 CH 3 ) 2 Rh 2 (Ac) 4 H CH(C 2 CH 3 ) 2 N 67% 97% % 1) CH 3 I 2) CH 3 CN reflux C 18 H 35 C 18 H 35 H NaH THF/H 2 71% C 18 H 35 C 18 H DCC 15 Me NH 2 n Me n H N ST152 (61%) ST155 (70%) 11
12 4. Detailed Description of Me- PEG- VE- DG Lipid Syntheses 1- (1,2 - Di- - oleyl- rac- glyceryloxy)propene (2). t- BuK (552 mg, 4.92 mmol) was added to a flask containing 1 (3.114 g, 4.92 mmol) in DMS (50 ml). The mixture was stirred at 100 C for 3 h and then diluted with diethyl ether (200 ml) before washing with water (2 x 50 ml). Silica gel column chromatographic purification (10:1 hexane:et 2 as eluent) of the organic residue gave 2 as an oil (2.881 g, 4.55 mmol, 93% yield). 1 H NMR (CDCl 3 ): δ 0.86 (t, 6H), 1.26 (m, 44H), 1.54 (m, 7H), 1.98 (m, 8H), (m, 9H), 4.35 (quintet, 1H, J = 6 Hz), 5.32 (m, 4H), 5.94 (d, 1H, J = 6 Hz); 13 C NMR (CDCl 3 ): δ 9.4, 14.3, 22.9, 26.3, 26.4, 27.5, 29.4, 29.5, 29.6, 29.7, 29.8, 29.9, 30.0, 30.3, 32.2, 32.8, 70.5, 71.0, 71.9, 72.4, 78.0, 101.1, 130.1, 130.2, 146.3; CI (M + H) + calc d 633, found (1,2 - Di- - oleyl- rac- glyceryloxy)- 1- (ω- methoxy- polyethylene glycol[2000]oxy)- 2- iodopropane (3). N- Iodosuccinimide (246 mg, 1.10 mmol) was added to a flask containing 2 (750 mg, 1.18 mmol) and mpeg2000 (1.82 g, mmol) in CH 2 Cl 2 (20 ml) at 0 C and the mixture stirred at 23 C for 3 h. The reaction mixture then was directly loaded onto a silica gel column and purified via step gradient elution with 20:1, then 10:1 CH 2 Cl 2 :MeH to give 3 (1.385 g, 5.02 mmol, 55%). 1 H NMR (CDCl 3 ): δ 0.85 (t, 6H, J = 6 Hz), 1.25 (m, 44H), 1.52 (m, 4H), 1.83 (d, 3H, J = 7 Hz), 1.98 (m, 8H), (m, ca. 200H), 4.16 (m, 1H), 4.45 (m, 1H), 5.32 (m, 4H). 1- (1,2 - Di- - oleyl- rac- glyceryloxy)- 1- (ω- methoxy- polyethylene glycol[5000]oxy)- 2- iodopropane (4). Compound 4 was prepared as described above in 41% yield using mpeg H NMR (CDCl 3 ): δ 0.85 (t, 6H, J = 6 Hz), 1.25 (m, 44H), 1.52 (m, 4H), 1.83 (d, 3H, J = 7 Hz), 1.98 (m, 8H), (m, ca. 500H), 4.16 (m, 1H), 4.45 (m, 1H), 5.32 (m, 4H). ST902. t- BuK (117 mg, 1.04 mmol) was added to 30 ml of THF containing 3 (1.308g, mmol) and the mixture stirred at 23 C for 4 h. The reaction mixture then was diluted with CH 2 Cl 2 (50 ml) and washed with 0.05 M of Na 2 C 3 solution (10 ml). The organic layer was dried over anhydrous 12
13 Na 2 C 3 and concentrated to give a solid. The resulting solid was dissolved in benzene and lyophilized to give ST902 (1.148 g, mmol, 92%). 1 H NMR (CDCl 3 ): δ 0.85 (t, 6H, J = 6 Hz), 1.25 (m, 44H), 1.51 (d, 3H, J = 7 Hz), 1.52 (m, 4H), 1.98 (m, 8H), (m, ca. 200H), 5.32 (m, 4H). ST905. ST905 was prepared from 4 in 69% yield as described above for ST H NMR (CDCl 3 ): δ 0.85 (t, 6H, J = 6 Hz), 1.25 (m, 44H), 1.51 (d, 3H, J = 7 Hz), 1.52 (m, 4H), 1.98 (m, 8H), (m, ca. 500H), 5.32 (m, 4H). 1,2- Di- - oleyl- rac- glycerol (5). t- BuK (1.514 g, 13.5 mmol) was added to a flask containing 1 (8.541 g, 13.5 mmol) in DMS (100 ml). The mixture was stirred at 100 C overnight, then diluted with hexane (300 ml) and washed with water (100 ml). The organic layer was dried over anhydrous MgS 4, concentrated under reduced pressure, and the resulting residue dissolved in a mixed solvent of MeH:CH 2 Cl 2 (50 ml:20 ml). p- TsH (300 mg, 1.58 mmol) was added and the mixture stirred at 23 C for 1 h before concentrating it to 20 ml and then adding 200 ml CH 2 Cl 2. The organic solution was washed with 0.05 M Na 2 C 3 solution (100 ml) and water (100 ml). Silica gel column chromatographic purification (2:1 hexane:et 2 as eluent) of the organic residue gave 5 as an oil (6.952 g, 11.7 mmol, 87% yield). 1 H NMR (CDCl 3 ): δ 0.86 (t, 6H), 1.26 (m, 44H), 1.54 (m, 4H), 1.98 (m, 8H), 2.1 (s, 1H), (m, 9H), 5.32 (m, 4H); 13 C NMR (CDCl 3 ): δ 14.3, 22.9, 26.3, 27.4, 29.5, 29.6, 29.7, 29.8, 29.9, 30.0, 30.3, 32.2, 32.8, 63.3, 70.6, 71.2, 72.1, 78.5, 130.1, 130.2; CI (M + H) + calc d 593, found (1,2 - Di- - oleyl- rac- glyceryloxy)ethene (6). Pd(Ac) 2 (217 mg, 0.97 mmol) and 1,10- phenanthroline (174 mg, 0.97 mmol) were added to a dry 100 ml Schlenk flask containing 5 (5.743 g, 9.68 mmol) in a mixture of ethyl vinyl ether (30 ml) and CH 2 Cl 2 (15mL) under an atmosphere of Ar. The mixture was stirred at 70 C for 20 h, prior to cooling to 0 C and exposure to air. The mixture was filtered using a short alumina column and the filtrate concentrated. Silica gel column chromatographic purification (8:1 hexane:et 2 as eluent) gave 6 as an oil (4.826 g, 8.80 mmol, 81% 13
14 yield) and recovered starting material 5 (1.098 g, 1.85 mmol, 19%). 1 H NMR (CDCl 3 ): δ 0.86 (t, 6H), 1.26 (m, 44H), 1.54 (m, 4H), 1.98 (m, 8H), (m, 9H), 3.97 (dd, 1H, J =2, 7 Hz), 4.17 (dd, 1H, J = 2, 14 Hz), 5.32 (m, 4H), 6.45 (dd, 1H, J = 7, 14 Hz); 13 C NMR (CDCl 3 ): δ 14.3, 22.9, 26.3, 26.4, 27.4, 29.4, 29.5, 29.6, 29.7, 29.8, 29.9, 30.0, 30.3, 32.2, 32.8, 68.3, 70.5, 70.9, 71.9, 77.0, 86.7, 130.0, 130.1, 152.2; CI (M + H) + calc d 619, found (1,2 - Di- - oleyl- rac- glyceryloxy)- 1- (ω- methoxy- polyethylene glycol[2000]oxy)- 2- iodoethane (7). Compound 7 was prepared in 44% yield starting from 6 and mpeg2000 using the same procedure described for the synthesis of 3. 1 H NMR (CDCl 3 ): δ 0.85 (t, 6H, J = 6 Hz), 1.25 (m, 44H), 1.51 (m, 4H), 1.97 (m, 8H), 3.21 (d, 2H, J = 5 Hz), (m, ca. 200H), 4.68 (m, 1H), 5.32 (m, 4H). 1- (1,2 - Di- - oleyl- rac- glyceryloxy)- 1- (ω- methoxy- polyethylene glycol[5000]oxy)- 2- iodoethane (8). Compound 8 was prepared in 52% yield starting from 6 and mpeg5000 using same procedure described for the synthesis of 3. 1 H NMR (CDCl 3 ): δ 0.85 (t, 6H, J = 6 Hz), 1.25 (m, 44H), 1.51 (m, 4H), 1.97 (m, 8H), 3.21 (d, 2H, J = 5 Hz), (m, ca. 500H), 4.68 (m, 1H), 5.32 (m, 4H). ST912. ST912 was prepared in 75% yield starting from 7 using the same procedure described for the synthesis of ST H NMR (CDCl 3 ): δ 0.84 (t, 6H, J = 6 Hz), 1.24 (m, 44H), 1.51 (m, 4H), 1.97 (m, 8H), 3.12 (s, 2H), (m, ca. 200H), 5.32 (m, 4H). ST915. ST915 was prepared in 94% yield starting from 8 using the same procedure described for the synthesis of ST H NMR (CDCl 3 ): δ 0.84 (t, 6H, J = 6 Hz), 1.24 (m, 44H), 1.51 (m, 4H), 1.97 (m, 8H), 3.12 (s, 2H), (m, ca. 500H), 5.32 (m, 4H). 1,2- Dioleyl- rac- 3- (2 - t- butyldimethylsilyloxy)acetate (9). xalyl chloride (1.02 ml, 11.7 mmol) was added to a flask containing bis(t- butyldimethylsilyloxy)glycolate (2.965g, 9.74 mmol) and 2 14
15 drops of DMF in CH 2 Cl 2 (15 ml) at 0 C. The mixture was stirred at 0 C for 10 min and then at 23 C for 2 h before concentrating the reaction mixture under reduced pressure and then dissolving the resulting residue with CH 2 Cl 2 (20 ml). Compound 5 (4.813 g, 8.12 mmol) in pyridine was added at 0 C and the reaction mixture stirred at 23 C for 2 h. The mixture then was diluted with CH 2 Cl 2 (50 ml) and washed with water (50 ml). Silica gel column chromatographic purification (8:1 hexane:et 2 as eluent) of the organic residue gave 9 as an oil (4.021 g, 5.25 mmol, 65% yield). 1 H NMR (CDCl 3 ): δ 0.08 (s, 6H), 0.87 (m, 15H), 1.24 (m, 44H), 1.53 (m, 4H), 1.98 (m, 8H), (m, 7H), (m, 4H), 5.32 (m, 4H); 13 C NMR (CDCl 3 ): δ - 5.2, 14.3, 18.6, 22.9, 26.0, 26.3, 27.4, 29.2, 29.3, 29.4, 29.5, 29.6, 29.7, 29.8, 29.9, 30.0, 30.3, 32.2, 32.8, 61.9, 64.5,70.4, 70.9, 72.0, 76.7, 130.0, 130.1, 171.8; ESI (M + H) + calc d 765, found ,2- Di- - oleyl- rac- glyceryloxy- 3- (1 - t- butyldimethylsilyloxy- 2 - propene) (10). TiCl 4 (2.14 ml, 19.5 mmol) was slowly added to THF (100 ml) at 0 C and the solution became yellow. TMEDA (5.9 ml, 39 mmol) was added at 0 C and the yellow solution became brown. Zn (2.868 mg, 43.8 mmol) containing 3% Pb was added and the solution stirred at 23 C for 30 min (the brown solution became green). A mixture of 9 (3.731, mmol) and CH 2 Br 2 in THF (10 ml) was added at 0 C and the mixture stirred at 23 C for 1 d. Saturated K 2 C 3 (10 ml) was added at 0 C and the mixture stirred at 0 C for 15 min before filtering the reaction mixture to remove the hydroxy metalate salts and concentrating the filtrate under reduced pressure. Alumina column chromatographic purification (2:1 hexane:ch 2 Cl 2 as eluent) of the organic residue gave 10 as an oil (1.543 g, 2.02 mmol, 42% yield). 1 H NMR (C 6 D 6 ): δ 0.04 (s, 6H), 0.87 (m, 15H), 1.24 (m, 44H), 1.55 (m, 4H), 2.06 (m, 8H), 3.33 (t, 2H, J = 7 Hz), (m, 4H), (m, 3H), 4.08 (s, 1H), 4.12 (s, 2H), 4.40 (s, 1H), 5.46 (m, 4H); 13 C NMR (C 6 D 6 ): δ - 5.2, 14.3, 18.5, 23.1, 26.1, 26.6, 27.7, 29.6, 29.7, 29.8, 29.9, 30.0, 30.1, 30.2, 30.7, 32.3, 33.1, 63.6, 68.2, 70.8, 71.3, 71.8, 77.6, 81.2, 130.2, 162.4; ESI (M + H) + calc d 763, found
16 1,2- Di- - oleyl- rac- glyceryloxy- 2 - propen- 1 - ol (11). TBAF (3.0 ml, 1.0 M in THF) was added to a flask containing 10 (763 mg, 1.00 mmol) in THF (10 ml). The mixture was stirred at 23 C for 2 h and filtered using a short alumina column (Et 2 eluent). The solution was concentrated and the residue purified via silica gel column chromatography (1:1 hexane:et 2 as eluent) to give 11 as an oil (594 mg, 0.92 mmol, 92% yield). 1 H NMR (CDCl 3 ): δ 0.85 (t, 6H, J = 7 Hz), 1.25 (m, 44H), 1.52 (m, 4H), 1.98 (m, 8H), 2.17 (s, 1H), 3.41 (t, 2H, J = 7 Hz), 3.49 (d, 2H, J = 7 Hz), 3.55 (t, 2H, J = 7 Hz), (m, 3H), 4.01 (m, 3H), 4.14 (s, 1H), 5.32 (m, 4H); 13 C NMR (CDCl 3 ): δ 14.1, 22.6, 26.0, 26.1, 27.2, 29.1, 29.2, 29.3, 29.4, 29.5, 29.6, 29.7, 29.8, 29.9, 30.0, 31.9, 32.6, 63.6, 67.6,70.4, 70.7, 71.7, 76.8, 82.3, 129.8, 129.9, 161.3; CI (M + H) + calc d 649, found ,2- Di- - oleyl- rac- glyceryl- 3- (2 - propenyl- 1 - (4 - nitrophenyl)carbonate) (12). Triethylamine (413 mg, 4.1 mmol) was added to 11 (531 mg, mmol) in THF (5 ml). 4- Nitrophenyl chloroformate (247 mg, 1.23 mmol) was added and the mixture stirred for 1 h. The reaction mixture was concentrated and the resulting residue purified by silica gel column chromatography (4:1 hexane:et 2 as eluent) to give 12 as an oil (612 mg, mmol, 92% yield). 1 H NMR (CDCl 3 ): δ 0.86 (t, 6H, J = 6 Hz), 1.24 (m, 44H), 1.53 (m, 4H), 1.97 (m, 8H), 3.43 (t, 2H, J = 7 Hz), 3.49 (d, 2H, J = 7 Hz), 3.56 (t, 2H, J = 7 Hz), (m, 3H), 4.21 (d, 1H, J = 3 Hz), 4.30 (d, 1H, J = 3 Hz), 4.67 (s, 2H), 5.31 (m, 4H), 7.37 (d, 2H, J = 9 Hz), 8.24 (d, 2H, J = 9 Hz); 13 C NMR (CDCl 3 ): δ 14.0, 22.6, 26.0, 27.2, 29.1, 29.2, 29.3, 29.4, 29.5, 29.6, 29.7, 30.0, 31.9, 32.5, 68.0, 68.9, 70.2, 70.8, 71.7, 76.7, 86.3, 121.7, 125.2, 129.7, 129.9, 145.4, 152.2, 155.5, 155.8; ESI calc d (M + Na) + 836, found 836. ST502. Triethylamine (230 mg, 2.3 mmol), 12 (380 mg, mmol), and mpegamine2000 (623 mg, mmol) were combined in DMF (10 ml) and stirred at 23 C for 12 h. The reaction mixture was concentrated and purified by silica gel column chromatography (10:1 CH 2 Cl 2 :MeH as eluent) to give ST502 (692 mg, mmol, 83% yield). 1 H NMR (CDCl 3 ): δ 0.80 (t, 6H, J = 6 Hz),
17 (m, 44H), 1.48 (m, 4H), 1.93 (m, 8H), (m, ca. 200H), 4.02 (d, 1H, J = 2 Hz), 4.10 (d, 1H, J = 2 Hz), 4.41 (s, 2H), 5.31 (m, 5H). ST505. ST505 was prepared as described above in 71% yield using mpegamine H NMR (CDCl 3 ): δ 0.82 (t, 6H, J = 6 Hz), 1.23 (m, 44H), 1.50 (m, 4H), 1.95 (m, 8H), (m, ca. 500H), 4.05 (d, 1H, J = 2 Hz), 4.13 (d, 1H, J = 2 Hz), 4.43 (s, 2H), 5.34 (m, 5H). Dimethyl 2- (1,2 - di- - oleyl- rac- glyceryloxy)malonate (13). Rh 2 (Ac) 4 (17 mg, mmol) was added to a flask containing 5 (2.234 g, 3.77 mmol) and dimethyl diazomalonate in benzene (50 ml). The mixture was heated at reflux for 3 h before cooling, concentrating the reaction mixture, and purifying the residue by silica gel column chromatography (1:1 hexane:et 2 as eluent) to give 13 as an oil (1.832 g, 2.53 mmol, 67% yield). 1 H NMR (CDCl 3 ): δ 0.85 (t, 6H, J = 7 Hz), 1.24 (m, 44H), 1.51 (m, 4H), 1.97 (m, 8H), (m, 9H), 3.78 (s, 6H), 4.65 (s, 1H), 5.31 (m, 4H); 13 C NMR (CDCl 3 ): δ 14.0, 22.6, 26.0, 26.1, 27.2, 29.1, 29.2, 29.3, 29.4, 29.5, 29.6, 29.7, 29.8, 29.9, 30.0, 31.5, 31.9, 32.6, 52.7, 70.0, 70.6, 71.5, 71.7, 77.9, 79.5, 129.8, 129.9, 166.9; ESI (M + H) + calc d 723, found 723. Dimethyl 2- (1,2 - di- - oleyl- rac- glyceryloxy)- 2- dimethylaminomethyl malonate (14). Eschenmoser s salt (1.454 g, 7.86 mmol) was added to a flask containing 13 (2.841 g, 3.93 mmol) and triethylamine (1.1 ml, 7.86 mmol) in CH 2 Cl 2 (50 ml). The mixture was stirred at 23 C for 12 h before washing the reaction mixture with water (2 x 20 ml) and concentration under reduced pressure. The resulting residue was purified by silica gel column chromatography (1:1 hexane:et 2 as eluent) to give 14 as an oil (2.970 g, 3.81 mmol, 97% yield). 1 H NMR (CDCl 3 ): δ 0.86 (t, 6H, J = 7 Hz), 1.25 (m, 44H), 1.52 (m, 4H), 1.98 (m, 8H), 2.28 (s, 6H), 2.91 (s, 2H), (m, 9H), 3.75 (s, 6H), 5.32 (m, 4H); 13 C NMR (CDCl 3 ): δ 14.0, 22.6, 26.0, 26.1, 27.2, 29.1, 29.2, 29.3, 29.4, 29.5, 29.6, 29.7, 30.0, 31.9, 32.5, 47.2, 52.3, 62.1, 66.7, 70.6, 70.9, 71.5, 77.7, 86.5, 129.8, 129.9, 168.3; ESI (M + H) + calc d 780, found
18 Methyl 2- (1,2 - di- - oleyl- rac- glyceryloxy)acrylate (15). MeI (1.36 ml, 21.8 mmol) was added to a flask containing 14 (3.01 g, 3.86 mmol) in CH 2 Cl 2 (50 ml). The mixture was heated at reflux for 12 h before cooling, concentration under reduced pressure and addition of CH 3 CN (50 ml). Then, the reaction mixture was heated at reflux for 1 d before concentration under reduced pressure, and purification of the organic residue by silica gel column chromatography (1:1 hexane:et 2 as eluent) to give 15 as an oil (2.012 g, 2.97 mmol, 99% yield). 1 H NMR (CDCl 3 ): δ 0.85 (t, 6H), 1.24 (m, 44H), 1.52 (m, 4H), 1.98 (m, 8H), (m, 12H), 4.62 (d, 1H, J = 2 Hz), 5.32 (m, 5H); 13 C NMR (CDCl 3 ): δ 14.0, 22.6, 26.0, 27.1, 29.1, 29.2, 29.3, 29.4, 29.5, 29.6, 29.7, 30.0, 31.8, 32.5, 52.1, 68.8, 70.2, 70.9, 71.7, 76.5, 94.3, 129.8, 129.9, 151.2, 163.4; ESI (M + H) + calc d 677, found (1,2 - Di- - oleyl- rac- glyceryloxy)acrylic acid (16). NaH solution (2.5 ml at 2.0 M) was added to a flask containing 15 (1.107 g, mmol) in a mixed solvent of THF (20 ml) and H 2 (10 ml). The reaction mixture was heated at reflux for 3 h and concentrated under reduced pressure. H 2 (20 ml) and Et 2 (20 ml) were added and the ph of the aqueous layer was adjusted to around 4 by slow addition of 1.0 M HCl. The organic layer was concentrated and the resulting residue purified by silica gel column chromatography (Et 2 as eluent) to give 16 as an oil (770 mg, 1.16 mmol, 71% yield). 1 H NMR (CDCl 3 ): δ 0.85 (t, 6H, J = 7 Hz), 1.25 (m, 44H), 1.54 (m, 4H), 1.98 (m, 8H), (m, 9H), 4.71 (d, 1H, J = 2 Hz), 5.31 (m, 4H), 5.46 (d, 1H, J = 2 Hz); 13 C NMR (CDCl 3 ): δ 14.0, 22.6, 26.0, 26.1, 27.2, 29.1, 29.2, 29.3, 29.4, 29.5, 29.6, 29.7, 30.0, 31.9, 32.5, 69.0, 70.0, 70.9, 71.8, 76.4, 76.0, 129.8, 129.9, 150.4, 166.2; ESI (M + H) + calc d 663, found 663. ST152. DCC (115 mg, mmol), 16 (493 mg, mmol), mpegamine2000 (743 mg, mmol), and DMAP (2 mg) were combined in CH 2 Cl 2 (30 ml) and stirred at 23 C for 12 h. The reaction mixture was filtered to remove the urea byproduct, the filtrate concentrated under reduced pressure, and the organic residue purified by silica gel column chromatography (10:1 CH 2 Cl 2 :MeH as 18
19 eluent) to give ST152 (603 mg, mmol, 61% yield). 1H NMR (CDCl3): δ 0.86 (t, 6H, J = 6 Hz), 1.25 (m, 44H), 1.54 (m, 4H), 1.98 (m, 8H), (m, ca. 200H), 4.44 (d, 1H, J = 2 Hz), 5.32 (m, 4H), 5.37 (d, 1H, J = 2 Hz), 6.94 (t, 1H, J = 5 Hz). ST155. ST155 was prepared as described above in 70% yield using mpegamine H NMR (CDCl3): δ 0.86 (t, 6H, J = 6 Hz), 1.25 (m, 44H), 1.54 (m, 4H), 1.98 (m, 8H), (m, ca. 500H), 4.44 (d, 1H, J = 2 Hz), 5.32 (m, 4H), 5.37 (d, 1H, J = 2 Hz), 6.94 (t, 1H, J = 5 Hz). 5. Hydrolysis Studies by TLC TLC"Analysis"of"mPEG2000<VE<Lipid"Hydrolysis" 0"hr" 3"hr" 6"hr" 9"hr" 12"hr" 18"hr" 24"hr" 48"hr" 72"hr" TLC"Analysis"of"mPEG5000<VE<Lipid"Hydrolysis" 0"hr" 3"hr" 6"hr" 9"hr" 12"hr" 19 18"hr" 24"hr" 48"hr"
20 6. Hydrolysis Studies by HPLC- CAD Figure S6. HPLC%Analysis%of%ST912%Hydrolysis% ST912,%t%=%0%hr% C 18 H 35 CC 15 H 31 H C 18 H 35 -PEG Me ST912% P 3 CH 2 CH 2 NMe 3 LPC% Standard% (1<PPC)% ST912,%t%=%48%hr% C 18 H 35 C 18 H 35 C 18 H 35 C 18 H 35 H Figure S7. HPLC%Analysis%of%ST502%Hydrolysis% CC 15 H 31 ST502,%t%=%0%hr% C 18 H 35 C 18 H 35 H P 3 CH 2 CH 2 NMe 3 LPC% Standard% (1<PPC)% NH-PEG Me ST502% ST502,%t%=%12%hr% C 18 H 35 C 18 H 35 H 20
21 Figure S8. HPLC%Analysis%of%ST302%Hydrolysis% ST302,%t%=%0%hr% C 18 H 35 C 18 H 35 CC 15 H 31 H P 3 CH 2 CH 2 NMe 3 LPC% Standard% (1=PPC)% NH-PEG Me ST302% ST302,%t%=%72%hr% C 18 H 35 C 18 H 35 H Figure S9. HPLC%Analysis%of%ST152%Hydrolysis% CC 15 H 31 ST152,%t%=%0%hr% C 18 H 35 C 18 H 35 H P 3 CH 2 CH 2 NMe 3 LPC% Standard% (1<PPC)% NH-PEG Me ST152% ST152,%t%=%72%hr% C 18 H 35 C 18 H 35 H 21
22 Figure S10. Table of Intrinsic Second rder Rate Constants and Proton Affinities Calculated by DFT mpeg-ve-lipid Pseudo-First rder Rate Constant (s-1) Intrinsic Second rder Rate Constant (M-1*s-1) log k (M -1 s -1 ) ST x x ST x ST x ST x x ST x x ST152 5 x x ST x x Figure S11. 1 H NMR of ST912 and ST902 hydrolysis proceeding in d 6 - benzene with <5% water added (ph 7). A B 0 h 0 h 12 h 12 h 36 h 36 h 22
23 % Calcein release A Calcein Release Rates for Various mpeg- VE- lipid Loadings in DPE Liposomes Figure S12. Release rates from DPE:ST152, DPE:ST155 and DPE:ST305 liposomes as a function of ph. mpeghn 5:95 ST152:DPE mpeg MW = 2000 C 18 H 35 C 18 H Time (min) ph 3.5 ph 4.5 ph 7.5 B % Calcein release C % Calcein release D % Calcein release mpeghn mpeg MW = :98 ST155:DPE C 18 H 35 C 18 H Time (min) 5:95 ST155:DPE Time (min) 8:92 ST155:DPE E % Calcein release Time (min) 2:98 ST305:DPE ph 3.5 ph 4.5 ph Time (min) ph 3.5 ph 4.5 ph 7.5 ph 3.5 ph 4.5 ph 7.5 ph 3.5 ph 4.5 ph
24 Figure S13. Calcein release rates from DPE:ST502 and DPE:ST505 liposomes as a function of ph. B % Calcein release :98 ST505:DPE mpeghn Time (min) mpeg MW = 5000 C 18 H 35 C 18 H 35 ph 3.5 ph 4.5 ph 7.5 % Calcein release A 5:95 ST502:DPE Time (min) ph 3.5 ph 4.5 ph 7.5 C % Calcein release 5:95 ST505:DPE Time (min) ph 3.5 ph 4.5 ph 7.5 D % Calcein release 8:92 ST505:DPE Time (min) ph 3.5 ph 4.5 ph
25 VI. References (1) Frisch, M. J.; Trucks, G. W.; Schlegel, H. B.; Scuseria, G. E.; Robb, M. A.; Cheeseman, J. R.; Montgomery, J., J.A.; Vreven, T.; Kudin, K. N.; Burant, J. C.; Millam, J. M.; Iyengar, S. S.; Tomasi, J.; Barone, V.; Mennucci, B.; Cossi, M.; Scalmani, G.; Rega, N.; Petersson, G. A.; Nakatsuji, H.; Hada, M.; Ehara, M.; Toyota, K.; Fukuda, R.; Hasegawa, J.; Ishida, M.; Nakajima, T.; Honda, Y.; Kitao,.; Nakai, H.; Klene, M.; Li, X.; Knox, J. E.; Hratchian, H. P.; Cross, J. B.; Adamo, C.; Jaramillo, J.; Gomperts, R.; Stratmann, R. E.; Yazyev,.; Austin, A. J.; Cammi, R.; Pomelli, C.; chterski, J. W.; Ayala, P. Y.; Morokuma, K.; Voth, G. A.; Salvador, P.; Dannenberg, J. J.; Zakrzewski, V. G.; Dapprich, S.; Daniels, A. D.; Strain, M. C.; Farkas,.; Malick, D. K.; Rabuck, A. D.; Raghavachari, K.; Foresman, J. B.; rtiz, J. V.; Cui, Q.; Baboul, A. G.; Clifford, S.; Cioslowski, J.; Stefanov, B. B.; Liu, G.; Liashenko, A.; Piskorz, P.; Komaromi, I.; Martin, R. L.; Fox, D. J.; Keith, T.; Al- Laham, M. A.; Peng, C. Y.; Nanayakkara, A.; Challacombe, M.; Gill, P. M. W.; Johnson, B.; Chen, W.; Wong, M. W.; Gonzalez, C.; Pople, J. A.; Gaussian 03, Gaussian, Inc.: Wallingford, CT, (2) Lee, C. T.; Yang, W. T.; Parr, R. G. Phys. Rev. B 1988, 37, 785. (3) Becke, A. D. J. Chem. Phys. 1993, 98, (4) Kendall, R. A.; Dunning, T. H. Jr.; Harrison, R. J. J. Chem. Phys. 1992, 96,
Supporting Information. Masahiro Egi, Kenji Azechi, Moriaki Saneto, Kaori Shimizu, and Shuji Akai*
 Supporting Information Cationic Gold(I)-Catalyzed Intramolecular Cyclization of -Hydroxyalkynones into 3(2H)-Furanones Masahiro Egi, Kenji Azechi, Moriaki Saneto, Kaori Shimizu, and Shuji Akai* School
Supporting Information Cationic Gold(I)-Catalyzed Intramolecular Cyclization of -Hydroxyalkynones into 3(2H)-Furanones Masahiro Egi, Kenji Azechi, Moriaki Saneto, Kaori Shimizu, and Shuji Akai* School
Hydrosulfide adducts of organo-iridium anticancer complexes
 Hydrosulfide adducts of organo-iridium anticancer complexes Pavel Štarha, Abraha Habtemariam, Isolda Romero-Canelo n, Guy J. Clarkson, Peter J. Sadler*, Regional Centre of Advanced Technologies and Materials,
Hydrosulfide adducts of organo-iridium anticancer complexes Pavel Štarha, Abraha Habtemariam, Isolda Romero-Canelo n, Guy J. Clarkson, Peter J. Sadler*, Regional Centre of Advanced Technologies and Materials,
Supporting Information
 Supporting Information Synthesis Of Novel Derivatives Of Thiophene- Carbazole, Their Electronic Properties And Computational Studies Damit E. F., 1 Nordin N., 1 Ariffin A.*,1 and Sulaiman K. 2 1 Chemistry
Supporting Information Synthesis Of Novel Derivatives Of Thiophene- Carbazole, Their Electronic Properties And Computational Studies Damit E. F., 1 Nordin N., 1 Ariffin A.*,1 and Sulaiman K. 2 1 Chemistry
POSS Lubricants and Blends
 PSS Lubricants and Blends usty L. Blanski, Shawn H. Phillips, Joe Lichtenhan, Joe Schwab, Andre Lee, Justin Leland, and Patrick uth AFL/PSM 10 E. Saturn Blvd. Edwards AFB CA 93524 661-525-5391 Edwards
PSS Lubricants and Blends usty L. Blanski, Shawn H. Phillips, Joe Lichtenhan, Joe Schwab, Andre Lee, Justin Leland, and Patrick uth AFL/PSM 10 E. Saturn Blvd. Edwards AFB CA 93524 661-525-5391 Edwards
******* 4 TH SECTION (PART I & II) *******
 ******* 4 TH SECTION (PART I & II) ******* Methods of increasing selectivity of is very unselective because it can form stable complexes with many divalent, trivalent and tetravalent cations. Many methods
******* 4 TH SECTION (PART I & II) ******* Methods of increasing selectivity of is very unselective because it can form stable complexes with many divalent, trivalent and tetravalent cations. Many methods
QUANTITATIVE MEASUREMENT OF FOUR MAJOR SECOIRIDOID DERIVATIVES IN OLIVE. OIL USING qnmr. PROOF OF THE ARTIFICIAL FORMATION OF ALDEHYDIC OLEUROPEIN AND
 QUANTITATIVE MEASUREMENT OF FOUR MAJOR SECOIRIDOID DERIVATIVES IN OLIVE OIL USING qnmr. PROOF OF THE ARTIFICIAL FORMATION OF ALDEHYDIC OLEUROPEIN AND LIGSTROSIDE AGLYCON ISOMERS EVANGELIA KARKOULA, ANGELIKI
QUANTITATIVE MEASUREMENT OF FOUR MAJOR SECOIRIDOID DERIVATIVES IN OLIVE OIL USING qnmr. PROOF OF THE ARTIFICIAL FORMATION OF ALDEHYDIC OLEUROPEIN AND LIGSTROSIDE AGLYCON ISOMERS EVANGELIA KARKOULA, ANGELIKI
Supporting Information for Achieving Accurate. Reduction Potential Predictions for. Anthraquinones in Water and Aprotic Solvents:
 Supporting Information for Achieving Accurate Reduction Potential Predictions for Anthraquinones in Water and Aprotic Solvents: Effects of Inter- and Intramolecular H-Bonding and Ion Pairing Hyungjun Kim,
Supporting Information for Achieving Accurate Reduction Potential Predictions for Anthraquinones in Water and Aprotic Solvents: Effects of Inter- and Intramolecular H-Bonding and Ion Pairing Hyungjun Kim,
Three years ( ) measurements of atmospheric concentrations of organochlorine pesticides (OCPs) at Station Nord, North East Greenland
 Three years (-) measurements of atmospheric concentrations of organochlorine pesticides (OCPs) at Station Nord, North East Greenland Rossana Bossi,* a Carsten Ambelas Skjøth,b and Henrik Skov a,c a Aarhus
Three years (-) measurements of atmospheric concentrations of organochlorine pesticides (OCPs) at Station Nord, North East Greenland Rossana Bossi,* a Carsten Ambelas Skjøth,b and Henrik Skov a,c a Aarhus
Temperature affects the silicate morphology in a diatom
 Supplementary Information: Temperature affects the silicate morphology in a diatom N. Javaheri 1, R. Dries 1&2, A. Burson 3, L.J. Stal 3&4, P.M.A. Sloot 1,5,6, J.A. Kaandorp 1* 1- Computational Science,
Supplementary Information: Temperature affects the silicate morphology in a diatom N. Javaheri 1, R. Dries 1&2, A. Burson 3, L.J. Stal 3&4, P.M.A. Sloot 1,5,6, J.A. Kaandorp 1* 1- Computational Science,
Vivaspin 500, 2, 6 and 20
 GE Healthcare Instructions 28-9331-02 AB Vivaspin 500, 2, 6 and 20 Intended use This product is intended for research use only, and shall not be used in any clinical or in vitro procedures for diagnostic
GE Healthcare Instructions 28-9331-02 AB Vivaspin 500, 2, 6 and 20 Intended use This product is intended for research use only, and shall not be used in any clinical or in vitro procedures for diagnostic
Supplementary Figure 1. HR-TEM images of unzipped carbon nanostructures by N- dopant-specific unzipping of NCNTs. a, Sequential unzipping of inner
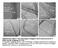 Supplementary Figure 1. HR-TEM images of unzipped carbon nanostructures by N- dopant-specific unzipping of NCNTs. a, Sequential unzipping of inner wall of NCNTs b,c, Unzipped nanostructures consisting
Supplementary Figure 1. HR-TEM images of unzipped carbon nanostructures by N- dopant-specific unzipping of NCNTs. a, Sequential unzipping of inner wall of NCNTs b,c, Unzipped nanostructures consisting
Determination of Pesticide Residues in Tea: An AOAC Collaborative Study
 Determination of Pesticide Residues in Tea: An AOAC Collaborative Study UCT Part Numbers: RFV5CT - 5 ml centrifuge tubes ECPSACB56-6 ml SPE cartridge with 5 mg GCB and 5 mg PSA ECSS25K - Sodium sulfate,
Determination of Pesticide Residues in Tea: An AOAC Collaborative Study UCT Part Numbers: RFV5CT - 5 ml centrifuge tubes ECPSACB56-6 ml SPE cartridge with 5 mg GCB and 5 mg PSA ECSS25K - Sodium sulfate,
SIMULTANEOUS RP HPLC METHOD DEVELOPMENT AND VALIDATION OF LEVOFLOXACIN AND ORNIDAZOLE IN COMBINED PHARMACEUTICAL DOSAGE FORMS
 Int. J. Chem. Sci.: 8(4), 2010, 2145-2152 SIMULTANEOUS RP HPLC METHOD DEVELOPMENT AND VALIDATION OF LEVOFLOXACIN AND ORNIDAZOLE IN COMBINED PHARMACEUTICAL DOSAGE FORMS CH. NARASIMHA RAJU BH *, K. V. RAMANA,
Int. J. Chem. Sci.: 8(4), 2010, 2145-2152 SIMULTANEOUS RP HPLC METHOD DEVELOPMENT AND VALIDATION OF LEVOFLOXACIN AND ORNIDAZOLE IN COMBINED PHARMACEUTICAL DOSAGE FORMS CH. NARASIMHA RAJU BH *, K. V. RAMANA,
GB Translated English of Chinese Standard: GB NATIONAL STANDARD OF THE
 Translated English of Chinese Standard: GB4789.3-2016 www.chinesestandard.net Sales@ChineseStandard.net GB NATIONAL STANDARD OF THE PEOPLE S REPUBLIC OF CHINA GB 4789.3-2016 National food safety standard
Translated English of Chinese Standard: GB4789.3-2016 www.chinesestandard.net Sales@ChineseStandard.net GB NATIONAL STANDARD OF THE PEOPLE S REPUBLIC OF CHINA GB 4789.3-2016 National food safety standard
Titan3 Syringe Filters. Sample Preparation 1-036
 Sample Preparation Titan3 Syringe Filters Low extractable membranes and housing HPLC performance tested Color coding for easy selection of the correct membrane and pore size Enhanced Luer Lock inlet which
Sample Preparation Titan3 Syringe Filters Low extractable membranes and housing HPLC performance tested Color coding for easy selection of the correct membrane and pore size Enhanced Luer Lock inlet which
Silica-based Solid Phase Extraction Columns
 Silica-based Solid Phase Extraction Columns Kinesis Ltd kinesis.co.uk Kinesis Inc kinesis-usa.com Kinesis GmbH (formerly Abimed) kinesisgmbh.de Kinesis Australia Pty Ltd kinesis-australia.com.au Contents
Silica-based Solid Phase Extraction Columns Kinesis Ltd kinesis.co.uk Kinesis Inc kinesis-usa.com Kinesis GmbH (formerly Abimed) kinesisgmbh.de Kinesis Australia Pty Ltd kinesis-australia.com.au Contents
TELOS Silica-based Solid Phase Extraction Columns From Kinesis
 TELOS Silica-based Solid Phase Extraction Columns From Kinesis Overview TELOS Non-polar (Reversed Phase) SPE Sorbents C18 C18(EC) C18 AQ(EC) C8(EC) C2(EC) TELOS Ion Exchange Sorbents SCX SAX NH2 TELOS
TELOS Silica-based Solid Phase Extraction Columns From Kinesis Overview TELOS Non-polar (Reversed Phase) SPE Sorbents C18 C18(EC) C18 AQ(EC) C8(EC) C2(EC) TELOS Ion Exchange Sorbents SCX SAX NH2 TELOS
Origin and Extraction of Andean Salars
 Think Simulation! Adventures in Electrolytes Origin and Extraction of Andean Salars Lithium and Boron Recovery from Salt brines and Salt flats in Chile and Bolivia http://tunari.tripod.com/landscapes.html
Think Simulation! Adventures in Electrolytes Origin and Extraction of Andean Salars Lithium and Boron Recovery from Salt brines and Salt flats in Chile and Bolivia http://tunari.tripod.com/landscapes.html
Diamond RealSeal Plastic Laboratory Bottles
 Diamond RealSeal Plastic Laboratory Bottles Premium bottles engineered to meet the requirements of the most demanding applications. Diamond RealSeal Plastic Laboratory Bottles Diamond RealSeal Plastic
Diamond RealSeal Plastic Laboratory Bottles Premium bottles engineered to meet the requirements of the most demanding applications. Diamond RealSeal Plastic Laboratory Bottles Diamond RealSeal Plastic
Bunsen burner and attached tubing, striker, evaporating dish, wire gauze pad, tongs.
 LABORATORY PROCEDURES OBJECTIVES: To observe proper safety techniques with all laboratory equipment. To use laboratory apparatus skillfully and efficiently. Before you actually complete an experiment,
LABORATORY PROCEDURES OBJECTIVES: To observe proper safety techniques with all laboratory equipment. To use laboratory apparatus skillfully and efficiently. Before you actually complete an experiment,
ISSN: Available online Journal of Global Trends in Pharmaceutical Sciences Vol.2, Issue 3, pp , July -Sept 2011
 Research Article ISSN: 2230-7346 Available online http://www.jgtps.com Journal of Global Trends in Pharmaceutical Sciences Vol.2, Issue 3, pp -264-276, July -Sept 2011 NEW RP-HPLC METHOD DEVELOPMENT AND
Research Article ISSN: 2230-7346 Available online http://www.jgtps.com Journal of Global Trends in Pharmaceutical Sciences Vol.2, Issue 3, pp -264-276, July -Sept 2011 NEW RP-HPLC METHOD DEVELOPMENT AND
9 LABORATORY ACCESSORY
 9 NORMAG - LABORATORY GLASSWARE L 309 e.1 DRYING TUBES They serve for drying of gas with suitable desiccants. To avoid sticking of the packing during the process, not shape resistant desiccants (e.g. phosphorus
9 NORMAG - LABORATORY GLASSWARE L 309 e.1 DRYING TUBES They serve for drying of gas with suitable desiccants. To avoid sticking of the packing during the process, not shape resistant desiccants (e.g. phosphorus
Investigation of the effect of antibiotics on bacterial growth. Introduction. Apparatus. Diagram of Apparatus
 Investigation of the effect of antibiotics on bacterial growth Introduction Antimicrobials are agents that are able to kill bacteria or halt their growth. They are widely used in medicine to treat bacterial
Investigation of the effect of antibiotics on bacterial growth Introduction Antimicrobials are agents that are able to kill bacteria or halt their growth. They are widely used in medicine to treat bacterial
Gas Chromatographic Presumptive Test for Coliform Bacteria in Water
 AmPID MICROBIOLOGY, Oct. 1975, P. 584-588 Copyright X) 1975 American Society for Microbiology Vol. 30, No. 4 Printed in U.SA. Gas Chromatographic Presumptive Test for Coliform Bacteria in Water JUDITH
AmPID MICROBIOLOGY, Oct. 1975, P. 584-588 Copyright X) 1975 American Society for Microbiology Vol. 30, No. 4 Printed in U.SA. Gas Chromatographic Presumptive Test for Coliform Bacteria in Water JUDITH
Silica-based Solid Phase Extraction Columns
 Silica-based Solid Phase Extraction Columns kinesis-group.com Contents TELOS Non-polar (Reversed Phase) SPE Sorbents Page 4 Page 5 Page 5 Page 6 Page 6 C18 C18(EC) C18 AQ(EC) C8(EC) C2(EC) TELOS Ion Exchange
Silica-based Solid Phase Extraction Columns kinesis-group.com Contents TELOS Non-polar (Reversed Phase) SPE Sorbents Page 4 Page 5 Page 5 Page 6 Page 6 C18 C18(EC) C18 AQ(EC) C8(EC) C2(EC) TELOS Ion Exchange
Structures of Solids. Prof Andrew Goodwin Michaelmas 2014
 Structures of Solids Prof Andrew Goodwin Michaelmas 2014 Li + Be 2+ N 3 O 2 F r/n ~ Polarisability Li + B C 76 22 48 70 133 Na + Mg 2+ Al 3+ Si 4+ P 3 S 2 Cl 102 36 18 10 70 92 181 K + Ca 2+ Sc 3+ Ti 3+
Structures of Solids Prof Andrew Goodwin Michaelmas 2014 Li + Be 2+ N 3 O 2 F r/n ~ Polarisability Li + B C 76 22 48 70 133 Na + Mg 2+ Al 3+ Si 4+ P 3 S 2 Cl 102 36 18 10 70 92 181 K + Ca 2+ Sc 3+ Ti 3+
OREGON Environmental Laboratory Accreditation Program ORELAP Fields of Accreditation BSK Associates Vancouver
 MATRIX Reference Code Analyte Code Description Drinking EPA 300.0 2.1 10053200 Methods for the Determination of Inorganic Substances in Environmental Samples 1575 Chloride 1730 Fluoride 1810 Nitrate as
MATRIX Reference Code Analyte Code Description Drinking EPA 300.0 2.1 10053200 Methods for the Determination of Inorganic Substances in Environmental Samples 1575 Chloride 1730 Fluoride 1810 Nitrate as
Coliform Count. Interpretation Guide. 3M Food Safety 3M Petrifilm Coliform Count Plate
 M Food Safety M Petrifilm Coliform Count Plate Coliform Count Interpretation Guide This guide familiarizes you with results on M Petrifilm Coliform Count Plates. For more information, contact the official
M Food Safety M Petrifilm Coliform Count Plate Coliform Count Interpretation Guide This guide familiarizes you with results on M Petrifilm Coliform Count Plates. For more information, contact the official
Safe - Spirals Protection for Hydraulic hoses
 Safeplast NA Company, Ltd. Safe - Spirals Protection for Hydraulic hoses Safeplast hose protectors provide safe and economic protection of hydraulic and pneumatic hoses thereby reducing machine downtime
Safeplast NA Company, Ltd. Safe - Spirals Protection for Hydraulic hoses Safeplast hose protectors provide safe and economic protection of hydraulic and pneumatic hoses thereby reducing machine downtime
Safe - Spirals Protection for Hydraulic hoses
 Safeplast NA Company, Ltd. Safe - Spirals Protection for Hydraulic hoses Safeplast hose protectors provide safe and economic protection of hydraulic and pneumatic hoses thereby reducing machine downtime
Safeplast NA Company, Ltd. Safe - Spirals Protection for Hydraulic hoses Safeplast hose protectors provide safe and economic protection of hydraulic and pneumatic hoses thereby reducing machine downtime
WÄRTSILÄ EXHAUST GAS CLEANING SYSTEM. Stian Aakre General Manager R&D. 1 Wärtsilä
 WÄRTSILÄ EXHAUST GAS CLEANING SYSTEM Stian Aakre General Manager R&D 1 Wärtsilä BPO Environmental Seminar 5 th Mach 2014 Wärtsilä scrubber system technology options Types and amounts of waste 2 Wärtsilä
WÄRTSILÄ EXHAUST GAS CLEANING SYSTEM Stian Aakre General Manager R&D 1 Wärtsilä BPO Environmental Seminar 5 th Mach 2014 Wärtsilä scrubber system technology options Types and amounts of waste 2 Wärtsilä
HARLECO Dyes & Stains Microscopy products to support your success
 HARLECO Dyes & Stains Microscopy products to support your success The life science business of Merck KGaA, Darmstadt, Germany operates as MilliporeSigma in the U.S. and Canada. A For over 90 years, HARLECO
HARLECO Dyes & Stains Microscopy products to support your success The life science business of Merck KGaA, Darmstadt, Germany operates as MilliporeSigma in the U.S. and Canada. A For over 90 years, HARLECO
HSCC. Interpretation Guide. High-Sensitivity Coliform Count Plate
 Interpretation Guide The 3M Petrifilm High-Sensitivity Coliform Count Plate is a sample-ready-culture medium system which contains modified Violet Red Bile (VRB) nutrients, cold-water-soluble gelling agent,
Interpretation Guide The 3M Petrifilm High-Sensitivity Coliform Count Plate is a sample-ready-culture medium system which contains modified Violet Red Bile (VRB) nutrients, cold-water-soluble gelling agent,
Geochemistry of Surface Water and Groundwater on the Campus of The Ohio State University. Senior Thesis
 Geochemistry of Surface Water and Groundwater on the Campus of The Ohio State University Senior Thesis Submitted in partial fulfillment of the requirements for the Bachelor of Science Degree, with Research
Geochemistry of Surface Water and Groundwater on the Campus of The Ohio State University Senior Thesis Submitted in partial fulfillment of the requirements for the Bachelor of Science Degree, with Research
Petrifilm. Interpretation Guide. Coliform Count Plate. Brand
 Petrifilm Brand Interpretation Guide The 3M Petrifilm is a sample-ready culture medium system that contains modified Violet Red Bile nutrients, a cold-water-soluble gelling agent and a tetrazolium indicator
Petrifilm Brand Interpretation Guide The 3M Petrifilm is a sample-ready culture medium system that contains modified Violet Red Bile nutrients, a cold-water-soluble gelling agent and a tetrazolium indicator
HARLECO Dyes & Stains Microscopy products to support your success
 HARLECO Dyes & Stains Microscopy products to support your success The life science business of Merck KGaA, Darmstadt, Germany operates as MilliporeSigma in the U.S. and Canada. A For over 90 years, HARLECO
HARLECO Dyes & Stains Microscopy products to support your success The life science business of Merck KGaA, Darmstadt, Germany operates as MilliporeSigma in the U.S. and Canada. A For over 90 years, HARLECO
Operating Instructions
 Operating Instructions High Throughput Dialysis MODEL HTD96b www.htdialysis.com HTD96b : Operating Instructions Components Universal Base Unit Teflon block assembly: 9 Teflon bars, labeled sequentially
Operating Instructions High Throughput Dialysis MODEL HTD96b www.htdialysis.com HTD96b : Operating Instructions Components Universal Base Unit Teflon block assembly: 9 Teflon bars, labeled sequentially
!"#$%&'()*+,-./012 ()*+,-. /0*+,123.
 !"#$%&'()*,.//0"34 (C) C a Ts(C ) 3Ts a!"#$%&'()*,./0 ()*,. /0*,3. 567(8(9%&'():;
!"#$%&'()*,.//0"34 (C) C a Ts(C ) 3Ts a!"#$%&'()*,./0 ()*,. /0*,3. 567(8(9%&'():;
Lab Equipment. Perry High School. Your lab equipment should:
 Lab Equipment Perry High School Your lab equipment should: 1. Be CLEAN before using it. 2. Be CHECKED (if glassware) for cracks, broken edges, and stars discard anything damaged. 3. Be washed, dried, and
Lab Equipment Perry High School Your lab equipment should: 1. Be CLEAN before using it. 2. Be CHECKED (if glassware) for cracks, broken edges, and stars discard anything damaged. 3. Be washed, dried, and
Nexus Standard Glove Box System
 NOW CONFORMS TO UL AND CSA STANDARDS FOR LABORATORY EQUIPMENT Nexus Standard Glove Box System Vacuum Atmospheres Company Vacuum Atmospheres Company 4652 W. Rosecrans Ave. East Coast Office P.O. Box 1043
NOW CONFORMS TO UL AND CSA STANDARDS FOR LABORATORY EQUIPMENT Nexus Standard Glove Box System Vacuum Atmospheres Company Vacuum Atmospheres Company 4652 W. Rosecrans Ave. East Coast Office P.O. Box 1043
Corning Aspirator Instruction Manual
 Corning Aspirator Instruction Manual Corning Aspirator Table of Contents Introduction.............................. 1 Set Up.................................... 1 Proper Use................................
Corning Aspirator Instruction Manual Corning Aspirator Table of Contents Introduction.............................. 1 Set Up.................................... 1 Proper Use................................
Old-Style Aging Cell 316 Stainless Steel mL Capacity. Part No Instruction Manual. Updated 8/21/2014 Ver. 1.7
 Old-Style Aging Cell 316 Stainless Steel - 500-mL Capacity Part No. 175-70 Instruction Manual Updated 8/21/2014 Ver. 1.7 OFI Testing Equipment, Inc. 11302 Steeplecrest Dr. Houston, Texas 77065 U.S.A. Tele:
Old-Style Aging Cell 316 Stainless Steel - 500-mL Capacity Part No. 175-70 Instruction Manual Updated 8/21/2014 Ver. 1.7 OFI Testing Equipment, Inc. 11302 Steeplecrest Dr. Houston, Texas 77065 U.S.A. Tele:
Comparison of Gelman and Millipore Membrane Filters for Enumerating Fecal Coliform Bacteria
 APPLIED MICROBIOLOGY, Sept. 1973, p. 332-336 Copyright 0 1973 American Society for Microbiology Vol. 26, No. 3 Printed in U.S.A. Comparison of Gelman and Millipore Membrane Filters for Enumerating Fecal
APPLIED MICROBIOLOGY, Sept. 1973, p. 332-336 Copyright 0 1973 American Society for Microbiology Vol. 26, No. 3 Printed in U.S.A. Comparison of Gelman and Millipore Membrane Filters for Enumerating Fecal
NOTES ON COST AND COST ESTIMATION by D. Gillen
 NOTES ON COST AND COST ESTIMATION by D. Gillen The basic unit of the cost analysis is the flight segment. In describing the carrier s cost we distinguish costs which vary by segment and those which vary
NOTES ON COST AND COST ESTIMATION by D. Gillen The basic unit of the cost analysis is the flight segment. In describing the carrier s cost we distinguish costs which vary by segment and those which vary
Minisart Syringe Filters
 Minisart Syringe Filters Removal of Particles and Microorganisms from Liquids and Gases Sample Preparation HPLC UHPLC Analytics Elimination of particles from your samples prior to HPLC or other chromatographic
Minisart Syringe Filters Removal of Particles and Microorganisms from Liquids and Gases Sample Preparation HPLC UHPLC Analytics Elimination of particles from your samples prior to HPLC or other chromatographic
Realax hoses & tubes specification. Fitted for the purpose
 Hoses pumps and tubes for APY, Series ISI, RP IP high and pressure RP series Realax hoses & tubes specification Hose pumps, also called peristaltic pumps, are excellent for moving a wide variety of fluids
Hoses pumps and tubes for APY, Series ISI, RP IP high and pressure RP series Realax hoses & tubes specification Hose pumps, also called peristaltic pumps, are excellent for moving a wide variety of fluids
Experimental Procedure. Lab 402
 Experimental Procedure Lab 402 A. Sample Preparation 1. Prepare a clean crucible Support the crucible and lid on a clay triangle and heat with an intense flame for 5 minutes. Clay triangle Allow them to
Experimental Procedure Lab 402 A. Sample Preparation 1. Prepare a clean crucible Support the crucible and lid on a clay triangle and heat with an intense flame for 5 minutes. Clay triangle Allow them to
Supplemental Table 1. TCRβ repertoire of naïve D b PA TRBV29 + CD8 + T cells in B6 mice. CDR3β M1 M2 M3 M4 M5 M6 SWGGEQ SWGERL 2 - -
 Supplemental Table 1. TCRβ repertoire of naïve D b PA + 224 TRBV29 + CD8 + T cells in B6 mice. CDR3β M1 M2 M3 M4 M5 M6 SWGGEQ 2-1 - 1 - SWGERL 2 - - - 1 - SPDRGAL 2 - - - - - SLGDEQ 1 - - - 1 1 AAGREQ
Supplemental Table 1. TCRβ repertoire of naïve D b PA + 224 TRBV29 + CD8 + T cells in B6 mice. CDR3β M1 M2 M3 M4 M5 M6 SWGGEQ 2-1 - 1 - SWGERL 2 - - - 1 - SPDRGAL 2 - - - - - SLGDEQ 1 - - - 1 1 AAGREQ
3M TM Petrifilm TM. Petrifilm TM 3M TM. 3M TM Petrifilm TM Serie 2000 Rapid Coliform Count Plates - Ref.: / 50 Unit - Ref.
 3M TM Aerobic Count Plates - Ref.: 06400 / 100 Unit - Ref.: 06406 / 1000 Unit 3M TM Enterobacteriaceae Count Plates 3M TM Coliform Count Plates - Ref.: 06420 / 50 Unit - Ref.: 06421 / 1000 Unit - Ref.:
3M TM Aerobic Count Plates - Ref.: 06400 / 100 Unit - Ref.: 06406 / 1000 Unit 3M TM Enterobacteriaceae Count Plates 3M TM Coliform Count Plates - Ref.: 06420 / 50 Unit - Ref.: 06421 / 1000 Unit - Ref.:
Gently apply pressure on spreader to distribute over circular area. Do not twist or slide the spreader. Interpretation
 0 With flat side down, place spreader on top film over inoculum. Gently apply pressure on spreader to distribute over circular area. Do not twist or slide the spreader. 2 Lift spreader. Wait at least one
0 With flat side down, place spreader on top film over inoculum. Gently apply pressure on spreader to distribute over circular area. Do not twist or slide the spreader. 2 Lift spreader. Wait at least one
Big SHOT III Hybridization Oven
 Big SHOT III Hybridization Oven Models 230402 and 230402-2 Whether you are working with Northern Blots, Southern Blots, Microarrays or another application that requires incubation, you will find the Big
Big SHOT III Hybridization Oven Models 230402 and 230402-2 Whether you are working with Northern Blots, Southern Blots, Microarrays or another application that requires incubation, you will find the Big
PACE APTITUDE & TALENT HUNT
 PACE APTITUDE & TALENT HUNT MUMBAI / NASHIK / PUNE / AKOLA / GOA / DELHI / LUCKNOW / KOLKATA ANSWERS KEY PAPER CODE-31 PATH TEST STUDENTS CURRENTLY STUDYING IN CLASS VI DATE: 04/09/2016 Q. No. Answer Q.
PACE APTITUDE & TALENT HUNT MUMBAI / NASHIK / PUNE / AKOLA / GOA / DELHI / LUCKNOW / KOLKATA ANSWERS KEY PAPER CODE-31 PATH TEST STUDENTS CURRENTLY STUDYING IN CLASS VI DATE: 04/09/2016 Q. No. Answer Q.
TOTAL COLIFORM ANDE.coli INDICATOR BACTERIA TEST KIT UV
 TOTAL COLIFORM ANDE.coli INDICATOR BACTERIA TEST KIT 4-3616-UV blank WARNING! This set contains chemicals that may be harmful if misused. Read cautions on individual containers carefully. Not to be used
TOTAL COLIFORM ANDE.coli INDICATOR BACTERIA TEST KIT 4-3616-UV blank WARNING! This set contains chemicals that may be harmful if misused. Read cautions on individual containers carefully. Not to be used
Ambient Temperature - 20 C to 80 C. 78mm W x 100mmL x 30mm H. Operating Voltage
 SSR Power Controller SSR 1 SSR 2 SSR 3 Phase Single 2 Leg Switching 3 Phase DIN Rail/Panel Mounting Both Both Both Cooling Fan Optional Optional Optional Control MethodZero Cross Trigger / high isolation
SSR Power Controller SSR 1 SSR 2 SSR 3 Phase Single 2 Leg Switching 3 Phase DIN Rail/Panel Mounting Both Both Both Cooling Fan Optional Optional Optional Control MethodZero Cross Trigger / high isolation
Chemical Resistance PPA 571
 Chemical Resistance PPA 571 - Prolonged contact refers to the process where the coating material is immersed in the chemical permanently, or intermittently for long periods of time. - Occasional short
Chemical Resistance PPA 571 - Prolonged contact refers to the process where the coating material is immersed in the chemical permanently, or intermittently for long periods of time. - Occasional short
Lab Equipment. Chemistry Willowridge High School Mr. L Moore
 Lab Equipment Chemistry Willowridge High School Mr. L Moore Lab Equipment Your lab equipment should: 1. Be CLEAN before using it. 2. Be CHECKED (if glassware) for cracks, broken edges, and stars discard
Lab Equipment Chemistry Willowridge High School Mr. L Moore Lab Equipment Your lab equipment should: 1. Be CLEAN before using it. 2. Be CHECKED (if glassware) for cracks, broken edges, and stars discard
chairs & stools-plastic V14b_Layout 1 11/07/ :05 Page 1 CHAIRS AND STOOLS - PLASTIC SHELL Primo Limited
 chairs & stools-plastic V14b_Layout 1 11/07/2018 10:05 Page 1 CHAIRS AND STOOLS - PLASTIC SHELL Primo Limited chairs & stools-plastic V14b_Layout 1 11/07/2018 10:05 Page 2 new P1918S 520mm 520mm 850mm
chairs & stools-plastic V14b_Layout 1 11/07/2018 10:05 Page 1 CHAIRS AND STOOLS - PLASTIC SHELL Primo Limited chairs & stools-plastic V14b_Layout 1 11/07/2018 10:05 Page 2 new P1918S 520mm 520mm 850mm
REC. Interpretation Guide. Rapid E. coli/coliform Count Plate
 Interpretation Guide The M Petrifilm Rapid E. coli/coliform Count Plate is a selective and differential sample-ready-culture medium system which contains proprietary nutrients, a cold-watersoluble gelling
Interpretation Guide The M Petrifilm Rapid E. coli/coliform Count Plate is a selective and differential sample-ready-culture medium system which contains proprietary nutrients, a cold-watersoluble gelling
Thermo Scientific Nalgene Rapid-Flow Filters. the last line of defense. against contamination
 Nalgene Rapid-Flow Filters the last line of defense against contamination The Rapid-Flow Advantage. From the outside, bottle top filters look alike. But, when you look beneath the membrane, glaring differences
Nalgene Rapid-Flow Filters the last line of defense against contamination The Rapid-Flow Advantage. From the outside, bottle top filters look alike. But, when you look beneath the membrane, glaring differences
Supplemental Information
 Neuron, Volume 88 Supplemental Information Time-Resolved Imaging Reveals Heterogeneous Landscapes of Nanomolar Ca 2+ in Neurons and Astroglia Kaiyu Zheng, Lucie Bard, James P. Reynolds, Claire King, Thomas
Neuron, Volume 88 Supplemental Information Time-Resolved Imaging Reveals Heterogeneous Landscapes of Nanomolar Ca 2+ in Neurons and Astroglia Kaiyu Zheng, Lucie Bard, James P. Reynolds, Claire King, Thomas
Water Filtration & Purification System
 INSTALLATION AND OPERATION INSTR UCTIONS FOR Series 200 High Flow Water Filtration & Purification System Table of Contents A. General Information B. Specifications C. Component Description D. Installation
INSTALLATION AND OPERATION INSTR UCTIONS FOR Series 200 High Flow Water Filtration & Purification System Table of Contents A. General Information B. Specifications C. Component Description D. Installation
3 Nomad Scraper Matting 6250 (formerly 3M Entrap All Seasons Matting)
 Technical Data/ Product Specifications 3 Nomad Scraper Matting 6250 (formerly 3M Entrap All Seasons Matting) 1.0 Product Description The product is a floor matting having a structure of vinyl filaments
Technical Data/ Product Specifications 3 Nomad Scraper Matting 6250 (formerly 3M Entrap All Seasons Matting) 1.0 Product Description The product is a floor matting having a structure of vinyl filaments
Pb 2+ Complete exchange 2 days. Zn 2+ Partial node exchange. Cd ncl2. 2 Cd 1.5(H3O)3[(Cd4O)3(HETT)8] 1 Zn Zn3[(Zn0.7Mn3.
![Pb 2+ Complete exchange 2 days. Zn 2+ Partial node exchange. Cd ncl2. 2 Cd 1.5(H3O)3[(Cd4O)3(HETT)8] 1 Zn Zn3[(Zn0.7Mn3. Pb 2+ Complete exchange 2 days. Zn 2+ Partial node exchange. Cd ncl2. 2 Cd 1.5(H3O)3[(Cd4O)3(HETT)8] 1 Zn Zn3[(Zn0.7Mn3.](/thumbs/94/121762313.jpg) Linker Original Linker Name L1 BTT = 1,3,5- benzenetristetrazolate L2 (HETT = 5,5,10,10,15,15 - Hexaethayltruxene- 2,7,12- MOF Original MOF Name Parent Metal Exchanged Metal Additional Notes Ref. 1 Mn
Linker Original Linker Name L1 BTT = 1,3,5- benzenetristetrazolate L2 (HETT = 5,5,10,10,15,15 - Hexaethayltruxene- 2,7,12- MOF Original MOF Name Parent Metal Exchanged Metal Additional Notes Ref. 1 Mn
Method development and validation for the simultaneous estimation of ambroxol HCL and levofloxacin by using RP HPLC method
 IJPAR Vol.6 Issue 4 Oct - Dec -2017 Journal Home page: ISSN:2320-2831 Research article Open Access Method development and validation for the simultaneous estimation of ambroxol HCL and levofloxacin by
IJPAR Vol.6 Issue 4 Oct - Dec -2017 Journal Home page: ISSN:2320-2831 Research article Open Access Method development and validation for the simultaneous estimation of ambroxol HCL and levofloxacin by
Texas Commission on Environmental Quality
 Method 40 CFR Part 50 Appendix L AB ID Method ID Fine particulates
Method 40 CFR Part 50 Appendix L AB ID Method ID Fine particulates
Silicon Wafers. Specifications. Front side Finish: Polished Back side Finish: Etched Primary Flat: Semi
 Silicon Wafers 2 inch p-type test wafers 2 inch n-type wafers Material: CZ Silicon Front side Finish: Polished Back side Finish: Etched Primary Flat: Semi Resistivity: min:1 to max:10 Ohm/cm N-Type Item#
Silicon Wafers 2 inch p-type test wafers 2 inch n-type wafers Material: CZ Silicon Front side Finish: Polished Back side Finish: Etched Primary Flat: Semi Resistivity: min:1 to max:10 Ohm/cm N-Type Item#
- Make sure the anaerobic tent will be available on the days you plan to use it. - Make sure the autoclave/incubator will be available.
 LytB Procedures - Make sure the anaerobic tent will be available on the days you plan to use it. - Make sure the autoclave/incubator will be available. - For the transformation of the host cells (E. coli
LytB Procedures - Make sure the anaerobic tent will be available on the days you plan to use it. - Make sure the autoclave/incubator will be available. - For the transformation of the host cells (E. coli
PM 2.5 source apportionment in the Po Valley: a 4 city study in Emilia-Romagna region
 PM 2.5 source apportionment in the Po Valley: a 4 city study in Emilia-Romagna region F. Scotto 1, I. Ricciardelli 1, D. Bacco 1, A. Vagheggini 2, A. Trentini 1, S. Ferrari 1, V. Poluzzi 1, C. Maccone
PM 2.5 source apportionment in the Po Valley: a 4 city study in Emilia-Romagna region F. Scotto 1, I. Ricciardelli 1, D. Bacco 1, A. Vagheggini 2, A. Trentini 1, S. Ferrari 1, V. Poluzzi 1, C. Maccone
ACQUITY Systems with 2D Technology
 ACQUITY Systems with 2D Technology MS Technology Day Sesión Alimentación y Medio Ambiente Barcelona, 21 de marzo de 2012 2012 Waters Corporation 1 Why Bother with 2D Chromatography? 2012 Waters Corporation
ACQUITY Systems with 2D Technology MS Technology Day Sesión Alimentación y Medio Ambiente Barcelona, 21 de marzo de 2012 2012 Waters Corporation 1 Why Bother with 2D Chromatography? 2012 Waters Corporation
Interpretation Guide. Coliform Count Plate
 Interpretation Guide The 3M Petrifilm is a sample-ready-culture medium system which contains modified Violet Red Bile nutrients, a cold-water-soluble gelling agent and a tetrazolium indicator that facilitates
Interpretation Guide The 3M Petrifilm is a sample-ready-culture medium system which contains modified Violet Red Bile nutrients, a cold-water-soluble gelling agent and a tetrazolium indicator that facilitates
GLOVE CONSTRUCTION SAFETY TERMS TERMS & CONDITIONS
 SAFETY TERMS TERMS & CONDITIONS TYPES OF LEATHER COW : Offers comfort, durability and breathability. Provides heat and abrasion resistance. PIG : Offers the greatest breathability due to its porous texture.
SAFETY TERMS TERMS & CONDITIONS TYPES OF LEATHER COW : Offers comfort, durability and breathability. Provides heat and abrasion resistance. PIG : Offers the greatest breathability due to its porous texture.
Premium Syringe Filters
 CapTiVa FilTraTioN Premium Syringe Filters More choices. Captiva syringe filters are available in a wide range of sizes, formats, and membranes to cover every matrix and sample. Certified. All products
CapTiVa FilTraTioN Premium Syringe Filters More choices. Captiva syringe filters are available in a wide range of sizes, formats, and membranes to cover every matrix and sample. Certified. All products
Interpretation Guide
 3M Petrifilm Interpretation Guide 3M Petrifilm Coliform Count Plates This guide familiarizes you with results on 3M Petrifilm Coliform Count Plates (CC). For further information, please contact the 3M
3M Petrifilm Interpretation Guide 3M Petrifilm Coliform Count Plates This guide familiarizes you with results on 3M Petrifilm Coliform Count Plates (CC). For further information, please contact the 3M
Interpretation Guide
 3M Petrifilm Interpretation Guide 3M Petrifilm Coliform Count Plates This guide familiarizes you with results on 3M Petrifilm Coliform Count Plates (CC). For further information, please contact the 3M
3M Petrifilm Interpretation Guide 3M Petrifilm Coliform Count Plates This guide familiarizes you with results on 3M Petrifilm Coliform Count Plates (CC). For further information, please contact the 3M
Gas Chromatograph Mass Spectrometer GCMS Consumables. p/n:
 Gas Chromatograph Mass Spectrometer GCMS Consumables p/n: 220-94898-01 Diag. # 1) Septum Nut 2) Needle Guide 3) Septum 4) Injection Port Assay 5) O-ring for Glass Insert 6) Glass Insert 7) Gold Gasket
Gas Chromatograph Mass Spectrometer GCMS Consumables p/n: 220-94898-01 Diag. # 1) Septum Nut 2) Needle Guide 3) Septum 4) Injection Port Assay 5) O-ring for Glass Insert 6) Glass Insert 7) Gold Gasket
Laboratories & Consulting Group
 Final Report Efficacy of Hypobromous Acid as a Hide Intervention Performed July 18-20, 2011 Submitted to Mike Harvey Technical Operations Mgr. Enviro Tech 500 Winmoore Way Modesto, CA 95358 209-232-2211
Final Report Efficacy of Hypobromous Acid as a Hide Intervention Performed July 18-20, 2011 Submitted to Mike Harvey Technical Operations Mgr. Enviro Tech 500 Winmoore Way Modesto, CA 95358 209-232-2211
Operating Instructions
 ID Self Pack Packing Device for ImmunoDetection Cartridges Operating Instructions In This Document This document describes how to pack your own ImmunoDetection (ID ) Sensor Cartridges for use in Perfusion
ID Self Pack Packing Device for ImmunoDetection Cartridges Operating Instructions In This Document This document describes how to pack your own ImmunoDetection (ID ) Sensor Cartridges for use in Perfusion
Microbial Hygiene Considerations with Mechanical Harvesting of Blueberries
 Microbial Hygiene Considerations with Mechanical Harvesting of lueberries Renée Allen UGA Extension, Alma, GA Dr. Harald Scherm UGA Plant Pathology, Athens, GA Dr. Jinru Chen UGA Food Science, Griffin,
Microbial Hygiene Considerations with Mechanical Harvesting of lueberries Renée Allen UGA Extension, Alma, GA Dr. Harald Scherm UGA Plant Pathology, Athens, GA Dr. Jinru Chen UGA Food Science, Griffin,
Addressing challenges associated with the detection of faecal coliform organisms in water matrices. Neil Leat Rand Water Date 30/09/2014
 Addressing challenges associated with the detection of faecal coliform organisms in water matrices Neil Leat Rand Water Date 30/09/2014 What are coliforms? Definitions of coliforms are based on biochemical
Addressing challenges associated with the detection of faecal coliform organisms in water matrices Neil Leat Rand Water Date 30/09/2014 What are coliforms? Definitions of coliforms are based on biochemical
ScienceDirect. Prediction of Commercial Aircraft Price using the COC & Aircraft Design Factors
 Available online at www.sciencedirect.com ScienceDirect Procedia Engineering 67 ( 2013 ) 70 77 7th Asian-Pacific Conference on Aerospace Technology and Science, 7th APCATS 2013 Prediction of Commercial
Available online at www.sciencedirect.com ScienceDirect Procedia Engineering 67 ( 2013 ) 70 77 7th Asian-Pacific Conference on Aerospace Technology and Science, 7th APCATS 2013 Prediction of Commercial
Testing Results of the Ecocina Cooking Stove from El Salvador By Nordica MacCarty March 5th, 2008
 Testing Results of the Cooking Stove from El Salvador By Nordica MacCarty March 5th, 28 Introduction The Stove, developed by Larry Winiarski and Nancy Hughes, was received at the Aprovecho laboratory in
Testing Results of the Cooking Stove from El Salvador By Nordica MacCarty March 5th, 28 Introduction The Stove, developed by Larry Winiarski and Nancy Hughes, was received at the Aprovecho laboratory in
data reports Ferrocenecarboxylic anhydride: a redetermination Structure description C. John McAdam and Jim Simpson*
 Ferrocenecarboxylic anhydride: a redetermination ISSN 2414-3146 C. John McAdam and Jim Simpson* Department of Chemistry, University of Otago, PO Box 56, Dunedin, New Zealand. *Correspondence e-mail: jsimpson@alkali.otago.ac.nz
Ferrocenecarboxylic anhydride: a redetermination ISSN 2414-3146 C. John McAdam and Jim Simpson* Department of Chemistry, University of Otago, PO Box 56, Dunedin, New Zealand. *Correspondence e-mail: jsimpson@alkali.otago.ac.nz
Carbon and Water Program Protocols for Neutrally Buoyant Sediment Traps Twilight Zone Explorer at BATS, June 2007-December 2009
 Carbon and Water Program Protocols for Neutrally Buoyant Sediment Traps Twilight Zone Explorer at BATS, June 2007-December 2009 MODIFIED ON OCTOBER 30, 2008 (S.OWENS); IN EFFECT NOVEMBER 2008 Wear gloves
Carbon and Water Program Protocols for Neutrally Buoyant Sediment Traps Twilight Zone Explorer at BATS, June 2007-December 2009 MODIFIED ON OCTOBER 30, 2008 (S.OWENS); IN EFFECT NOVEMBER 2008 Wear gloves
Composite CN Value Based on Hydrologic Soil Group and Estimated Future Land Cover
 Composite CN Value Based on Hydrologic Soil Group and Estimated Future Land Cover Village of Milan, Mirabal Park /5/016 DEVELOPED LAND AREAS** Basin Basin HSG "B" HSG "C" HSG "D" Basin Basin Area Area
Composite CN Value Based on Hydrologic Soil Group and Estimated Future Land Cover Village of Milan, Mirabal Park /5/016 DEVELOPED LAND AREAS** Basin Basin HSG "B" HSG "C" HSG "D" Basin Basin Area Area
NOTICE TO MEMBERS No February 5, 2003
 NOTICE TO MEMBERS No. 2003-008 February 5, 2003 NEW EQUITY OPTION CLASSES Bourse de Montréal Inc.(The Bourse) and (CDCC) hereby inform you that at the opening of trading on Monday, February 10, 2003 the
NOTICE TO MEMBERS No. 2003-008 February 5, 2003 NEW EQUITY OPTION CLASSES Bourse de Montréal Inc.(The Bourse) and (CDCC) hereby inform you that at the opening of trading on Monday, February 10, 2003 the
Interpretation Guide 3M Petrifilm Rapid Coliform Count Plates
 3M Petrifilm Interpretation Guide 3M Petrifilm Rapid Coliform Count Plates This guide should familiarize you with results on Petrifilm Rapid Coliform Count (RCC) plates as defined by three of the most
3M Petrifilm Interpretation Guide 3M Petrifilm Rapid Coliform Count Plates This guide should familiarize you with results on Petrifilm Rapid Coliform Count (RCC) plates as defined by three of the most
Installing the Agilent 6890 Oven Insert for Fast Chromatography
 Installing the Agilent 6890 Oven Insert for Fast Chromatography Kit G2646-60500 The 6890 Oven Insert for Fast Chromatography reduces the oven volume so that the column and sample heat more quickly, yielding
Installing the Agilent 6890 Oven Insert for Fast Chromatography Kit G2646-60500 The 6890 Oven Insert for Fast Chromatography reduces the oven volume so that the column and sample heat more quickly, yielding
Evaluation copy. Fecal Coliform. Computer INTRODUCTION
 Fecal Coliform Computer 9 INTRODUCTION The concentration of fecal coliform bacteria in water is measured to determine the likelihood of contamination by microbiological organisms. While fecal coliform
Fecal Coliform Computer 9 INTRODUCTION The concentration of fecal coliform bacteria in water is measured to determine the likelihood of contamination by microbiological organisms. While fecal coliform
Toyostove Error Code EE 2 (EE 6)
 Toyostove Error Code EE 2 (EE 6) WARNING Rural Energy Enterprises, Inc. does not accept liability for the improper use of this information. Installation, service, and maintenance of heating equipment should
Toyostove Error Code EE 2 (EE 6) WARNING Rural Energy Enterprises, Inc. does not accept liability for the improper use of this information. Installation, service, and maintenance of heating equipment should
Jonathan Howarth Ph.D and Tina Rodrigues BS Enviro Tech Chemical Services Modesto, CA 95258
 The Effectiveness of Water, Sodium Hypochlorite Bleach, and Peroxyacetic Acid (PAA) in Eradicating a Wild Field Strain of E. coli O157:H7 from the Surface of Cucumbers Background Jonathan Howarth Ph.D
The Effectiveness of Water, Sodium Hypochlorite Bleach, and Peroxyacetic Acid (PAA) in Eradicating a Wild Field Strain of E. coli O157:H7 from the Surface of Cucumbers Background Jonathan Howarth Ph.D
CENTRAL MARKS DEPARTMENT-III
 CENTRAL MARKS DEPARTMENT-III DOC No. CMD-III/IS 4246/1 Date : 24-01-2008 RECOMMENDED LIST OF TEST EQUIPMENT FOR DOMESTIC GAS STOVES FOR USE WITH LPG AS PER IS Clause Requirement as No. per 5. Materials
CENTRAL MARKS DEPARTMENT-III DOC No. CMD-III/IS 4246/1 Date : 24-01-2008 RECOMMENDED LIST OF TEST EQUIPMENT FOR DOMESTIC GAS STOVES FOR USE WITH LPG AS PER IS Clause Requirement as No. per 5. Materials
PIONEERING DRYING TECHNOLOGY
 SEPARATION PIONEERING DRYING TECHNOLOGY FLUIDIZED BED DRYING AND COOLING FOR THE SODA ASH INDUSTRY Pioneering drying technology for soda ash plants As the leader in technology development for efficient
SEPARATION PIONEERING DRYING TECHNOLOGY FLUIDIZED BED DRYING AND COOLING FOR THE SODA ASH INDUSTRY Pioneering drying technology for soda ash plants As the leader in technology development for efficient
Managing Hypochlorite to Reduce Chlorate Formation: A Utility Case Study
 Managing Hypochlorite to Reduce Chlorate Formation: A Utility Case Study KIMBERLY GUPTA, PE PORTLAND WATER BUREAU CO- AUTHOR: YONE AKAGI, PE AWWA PNWS KENNEWICK, MAY 5, 2017 Presentation Outline Background
Managing Hypochlorite to Reduce Chlorate Formation: A Utility Case Study KIMBERLY GUPTA, PE PORTLAND WATER BUREAU CO- AUTHOR: YONE AKAGI, PE AWWA PNWS KENNEWICK, MAY 5, 2017 Presentation Outline Background
Community College of Philadelphia. Department of Chemistry
 Community College of Philadelphia Department of Chemistry SAFETY TEST ANSWER KEY 1) Who should be immediately called for assistance in case of an accident in the laboratory? Your laboratory instructor.
Community College of Philadelphia Department of Chemistry SAFETY TEST ANSWER KEY 1) Who should be immediately called for assistance in case of an accident in the laboratory? Your laboratory instructor.
Confirmation Protocol for E. coli O157:H7
 Introduction Confirmation Protocol for E. coli O157:H7 The following protocol is used by Hygiena to recover E. coli O157:H7 from beef samples that were enriched according to the BAX System method. The
Introduction Confirmation Protocol for E. coli O157:H7 The following protocol is used by Hygiena to recover E. coli O157:H7 from beef samples that were enriched according to the BAX System method. The
Load-following capabilities of Nuclear Power Plants. Erik Nonbøl
 Load-following capabilities of Nuclear Power Plants Erik Nonbøl Outline Why load-following Modes of power operation BWR technique for load-following PWR technique for load-following Effects on components
Load-following capabilities of Nuclear Power Plants Erik Nonbøl Outline Why load-following Modes of power operation BWR technique for load-following PWR technique for load-following Effects on components
Why measure average air speed in a tunnel ventilated house?
 How To... A Tunnel Ventilated House Why measure average air speed in a tunnel ventilated house? The speed that air moves down the length of a poultry house determines the level of cooling that the birds
How To... A Tunnel Ventilated House Why measure average air speed in a tunnel ventilated house? The speed that air moves down the length of a poultry house determines the level of cooling that the birds
Corrosion resistance chart
 Corrosion resistance chart Date: 06/10 Korrosionstabelle Page 1 of 6 Acetaldehyd 40 40 NR NR NR Acetic Acid 10% to 50% 80 80 38 38 100 Acetic Acid 50% to 75% 65 65 24 24 75 Acetic Acid 75% to 100% 45 45
Corrosion resistance chart Date: 06/10 Korrosionstabelle Page 1 of 6 Acetaldehyd 40 40 NR NR NR Acetic Acid 10% to 50% 80 80 38 38 100 Acetic Acid 50% to 75% 65 65 24 24 75 Acetic Acid 75% to 100% 45 45
CHAPTER 8 SOLUTIONS TO EXERCISES
 CHAPTER 8 SOLUTIONS TO EXERCISES Solution 8.2. The walk starts with the anomeric proton (peak h), which can be unambiguously assigned because it is downfield of all the other peaks, and it is the only
CHAPTER 8 SOLUTIONS TO EXERCISES Solution 8.2. The walk starts with the anomeric proton (peak h), which can be unambiguously assigned because it is downfield of all the other peaks, and it is the only
SQUARE JARS Jar 250gm / 200ml Amber Square Pallet (3648 per pallet) 57mm neck Jar 250gm / 200ml Amber Square Carton (336 per carton) 57mm neck
 Mossop s Honey Date: 3 rd May 2016 761 State Highway 29 Tauriko RD1, Tauranga 3171 Attention: Wendy Mossop Dear Wendy, This is to confirm that products for Mossop s Honey as listed are manufactured as
Mossop s Honey Date: 3 rd May 2016 761 State Highway 29 Tauriko RD1, Tauranga 3171 Attention: Wendy Mossop Dear Wendy, This is to confirm that products for Mossop s Honey as listed are manufactured as
