Supporting Information. Masahiro Egi, Kenji Azechi, Moriaki Saneto, Kaori Shimizu, and Shuji Akai*
|
|
|
- Nancy May
- 5 years ago
- Views:
Transcription
1 Supporting Information Cationic Gold(I)-Catalyzed Intramolecular Cyclization of -Hydroxyalkynones into 3(2H)-Furanones Masahiro Egi, Kenji Azechi, Moriaki Saneto, Kaori Shimizu, and Shuji Akai* School of Pharmaceutical Sciences, University of Shizuoka, 52-1, Yada, Suruga-ku, Shizuoka, Shizuoka , Japan Contents Experimental Section General procedure for the preparation of 3(2H)-furanones and 2,3-dihydro-4H-pyran- S2 4-ones Spectroscopic data for 3(2H)-furanones 2a i S2 S3 Spectroscopic data for 2,3-dihydro-4H-pyran-4-ones 4a,b S4 Preparation of γ-hydroxyalkynones 1a i S4 S6 Preparation of δ-hydroxyalkynones 3a,b S7 1 H and 13 C NMR chart for 3(2H)-furanones 2a i S8 S25 1 H and 13 C NMR chart for 2,3-dihydro-4H-pyran-4-ones 4a,b S26 S29 1 H and 13 C NMR chart for γ-hydroxyalkynones 1a i S30 S47 1 H and 13 C NMR chart for δ-hydroxyalkynones 3a,b S48 S51 S1
2 General procedure for the preparation of 3(2H)-furanones and 2,3-dihydro-4H-pyran-4-ones To a solution of γ-hydroxyalkynones 1 or δ-hydroxyalkynones 3 (0.50 mmol) in toluene (2.5 ml, 0.20 M) were added (p-cf 3 C 6 H 4 ) 3 PAuCl (17.5 mg, mmol) and AgOTf (6.4 mg, mmol) in this order at room temperature. The reaction mixture was stirred at room temperature until the complete consumption of 1 and then quenched with sat. aq. NH 4 Cl. The organic materials were extracted with EtOAc, and the combined organic extracts were washed with brine, dried over MgSO 4, and evaporated in vacuo. The residue was purified by column chromatography (silica gel, usually hexanes/etoac) to give 3(2H)-furanone 2 and 2,3-dihydro- 4H-pyran-4-ones 4. Spectroscopic data for 3(2H)-furanones 2-Methyl-5-phenylethyl-3(2H)-furanone (2a) A pale yellow oil; 1 H NMR (500 MHz, CDCl 3 ) δ 1.43 (3H, d, J = 7.3 Hz), 2.82 (2H, t, J = 7.5 Hz), 2.98 (2H, t, J = 7.5 Hz), 4.48 (1H, q, J = 7.3 Hz), 5.39 (1H, s), (5H, m); 13 C NMR (125 MHz, CDCl 3 ) δ 16.3, 32.0, 32.4, 82.5, 103.1, 126.6, 128.2, 128.6, 139.6, 192.3, 205.6; IR (CHCl 3 ) ν 3015, 2935, 1695, 1590 cm 1. HRMS (FAB) m/z calcd for C 13 H 14 O 2 [M] + : Found: ,2-Dimethyl-5-phenylethyl-3(2H)-furanone (2b) 1 A pale yellow solid, mp C; 1 H NMR (500 MHz, CDCl 3 ) δ 1.35 (6H, s), 2.81 (2H, t, J = 8.0 Hz), 2.97 (2H, t, J = 8.0 Hz), 5.32 (1H, s), (5H, m); 13 C NMR (125 MHz, CDCl 3 ) δ 22.8, 32.1, 32.4, 88.6, 101.6, 126.6, 128.3, 128.6, 139.6, 190.4, 207.4; IR (CHCl 3 ) ν 3015, 2985, 2930, 1690, 1590 cm 1. 2,5-Dimethyl-2-phenyl-3(2H)-furanone (2c) 2 A pale yellow oil; 1 H NMR (500 MHz, CDCl 3 ) δ 1.74 (3H, s), 2.33 (3H, s), 5.40 (1H, s), (3H, m), (2H, m); 13 C NMR (125 MHz, CDCl 3 ) δ 16.9, 24.3, 90.4, 102.2, 124.5, 128.0, 128.5, 138.1, 188.5, 204.8; IR (CHCl 3 ) ν 3065, 3020, 1695, 1610 cm 1. 2,2-Dimethyl-5-(1,1-dimethylethyl)-3(2H)-furanone (2d) 3 A colorless solid, mp C; 1 H NMR (500 MHz, CDCl 3 ) δ 1.23 (9H, s), 1.35 (6H, s), 5.32 (1H, s); 13 C NMR (125 MHz, CDCl 3 ) δ 22.8, 27.4, 34.5, 88.4, 98.0, 199.0, 208.0; IR (CHCl 3 ) ν 3010, 2980, 1690, 1575 cm 1. 1 Reiter, M.; Turner, H.; Mills-Webb, R.; Gouverneur, V. J. Org. Chem. 2005, 70, Chimichi, S.; Boccalini, M.; Cosimelli, B.; Dall Acqua, F.; Viola, G. Tetrahedron 2003, 59, Sampson, P.; Roussis, V.; Drtina, G. J.; Koerwitz, F. L.; Wiemer, D. F. J. Org. Chem. 1986, 51, S2
3 2-Pentyl-1-oxaspiro[4,4]non-2-en-4-one (2e) 4 A yellow oil; 1 H NMR (500 MHz, CDCl 3 ) δ 0.90 (3H, t, J = 7.2 Hz), (4H, m), (2H, m), (8H, m), 2.45 (2H, t, J = 7.4 Hz), 5.38 (1H, s); 13 C NMR (125 MHz, CDCl 3 ) δ 13.9, 22.2, 25.6, 25.7, 30.8, 31.2, 36.9, 98.5, 102.3, 192.3, 206.6; IR (CHCl 3 ) ν 2960, 2935, 1680, 1585 cm 1. HRMS (ESI) m/z calcd for C 13 H 20 O 2 Na [M+Na] + : Found: ,2-Dimethyl-5-phenyl-3(2H)-furanone (2f) 1 An off-white solid, mp C; 1 H NMR (500 MHz, CDCl 3 ) δ 1.49 (6H, s), 5.97 (1H, s), (3H, m), 7.84 (2H, d, J = 8.0 Hz); 13 C NMR (125 MHz, CDCl 3 ) δ 23.1, 89.0, 98.6, 127.2, 128.8, 129.2, 132.6, 183.4, 207.0; IR (CHCl 3 ) ν 3015, 2985, 1685, 1605 cm 1. 5-(4-Methoxyphenyl)-2,2-dimethyl-3(2H)-furanone (2g) 1 A pale yellow solid, mp C; 1 H NMR (500 MHz, CDCl 3 ) δ 1.48 (6H, s), 3.88 (3H, s), 5.85 (1H, s), 6.98 (2H, d, J = 8.8 Hz), 7.78 (2H, d, J = 8.8 Hz); 13 C NMR (125 MHz, CDCl 3 ) δ 23.2, 55.5, 88.8, 97.0, 114.2, 121.7, 129.1, 163.2, 183.3, 206.7; IR (CHCl 3 ) ν 3020, 2985, 1680, 1610 cm 1. (E)-2,2-Dimethyl-5-(2-phenylethenyl)-3(2H)-furanone (2h) 5 An orange solid, mp C; 1 H NMR (500 MHz, CDCl 3 ) δ 1.46 (6H, s), 5.59 (1H, s), 6.86 (1H, d, J = 16.0 Hz), (3H, m), 7.50 (1H, d, J = 16.0 Hz), (2H, m); 13 C NMR (125 MHz, CDCl 3 ) δ 23.2, 88.5, 102.0, 116.4, 127.9, 129.0, 130.2, 134.7, 138.8, 181.3, 206.9; IR (CHCl 3 ) ν 3015, 2985, 1680, 1630 cm 1. 5-Phenylethyl-3(2H)-furanone (2i) A pale yellow oil; H NMR (500 MHz, CDCl 3 ) δ 2.84 (2H, t, J = 8.0 Hz), 2.98 (2H, t, J = 8.0 Hz), 4.50 (2H, s), 5.47 (1H, s), (5H, m); 13 C NMR (125 MHz, CDCl 3 ) δ 31.9, 32.4, 75.2, 104.5, 126.6, 128.2, 128.7, 139.6, 194.0, 202.9; IR (CHCl 3 ) ν 3015, 2965, 1695, 1595 cm 1. HRMS (ESI) m/z calcd for C 12 H 12 O 2 Na [M+Na] + : Found: Binder, J. T.; Crone, B.; Kirsch, S. F.; Liébert, C.; Menz, H. Eur. J. Org. Chem. 2007, Inoue, Y.; Ohuchi, K.; Yen, I.-F.; Imaizumi, S. Bull. Chem. Soc. Jpn. 1989, 62, S3
4 Spectroscopic data for 2,3-dihydro-4H-pyran-4-ones 6-Phenyl-2,3-dihydro-4H-pyran-4-one (4a) A pale yellow solid, mp C; 1 H NMR (500 MHz, CDCl 3 ) δ 2.67 (2H, t, J = 7.0 Hz), 4.67 (2H, t, J = 7.0 Hz), 6.03 (1H, s), (3H, m), 7.74 (2H, d, J = 7.5 Hz); 13 C NMR (125 MHz, CDCl 3 ) δ 36.0, 68.2, 102.4, 126.5, 128.7, 131.7, 132.6, 170.5, 192.7; IR (CHCl 3 ) ν 3020, 1660, 1600 cm 1. HRMS (ESI) m/z calcd for C 11 H 10 O 2 Na [M+Na] + : Found: Phenylethyl-2,3-dihydro-4H-pyran-4-one (4b) A pale yellow oil; 1 H NMR (500 MHz, CDCl 3 ) δ 2.51 (2H, t, J = 6.9 Hz), 2.55 (2H, t, J = 7.7 Hz), 2.88 (2H, t, J = 7.7 Hz), 4.45 (2H, t, J = 6.8 Hz), 5.30 (1H, s), (5H, m); 13 C NMR (125 MHz, CDCl 3 ) δ 32.5, 35.7, 36.5, 68.0, 105.0, 126.4, 128.2, 128.5, 140.1, 176.5, 192.1; IR (CHCl 3 ) ν 3015, , 1605 cm 1. HRMS (ESI) m/z calcd for C 13 H 14 O 2 Na [M+Na] + : Found: General procedure for the preparation of γ-hydroxyalkynones To a solution of propargyl alcohols (9.0 mmol) in THF (15 ml) were added HMPA (36 mmol) and n-butyllithium (1.6 M in hexanes, 18 mmol) sequentially at 78 C under nitrogen atmosphere. After stirring for 30 min at 78 C, a solution of Weinreb amides (7.5 mmol) in THF (5.0 ml) was added. The reaction mixture was allowed to warm to room temperature, stirred until the complete consumption of Weinreb amides, and quenched with sat. aq. NH 4 Cl. The organic materials were extracted with EtOAc, and the combined organic extracts were washed with H 2 O several times and then brine, dried over MgSO 4, and evaporated in vacuo. The residue was purified by column chromatography (silica gel, usually hexanes/etoac) to give 1. Spectroscopic data for γ-hydroxyalkynones 6-Hydroxy-1-phenyl-4-heptyn-3-one (1a) 36% yield: Prepared from N-methoxy-N-methyl-3-phenylpropionamide and 3-butyn-2-ol. A pale yellow oil; 1 H NMR (500 MHz, CDCl 3 ) δ 1.51 (3H, d, J = 6.3 Hz), 1.95 (1H, d, J = 5.7 Hz), (4H, m), (1H, m), (5H, m); 13 C NMR (125 MHz, CDCl 3 ) δ 23.4, 29.7, 46.7, 58.1, 82.7, 93.0, 126.3, 128.3, 128.5, 140.0, 186.6; IR (CHCl 3 ) ν 3600, 3445, 3015, 2215, 1675 cm 1. HRMS (ESI) m/z calcd for C 13 H 14 O 2 Na [M+Na] + : Found: S4
5 6-Hydroxy-6-methyl-1-phenyl-4-heptyn-3-one (1b) 72% yield: Prepared from N-methoxy-N-methyl-3-phenylpropionamide and 2-methyl-3-butyn-2-ol. A pale yellow oil; 1 H NMR (500 MHz, CDCl 3 ) δ 1.55 (6H, s), 2.10 (1H, br s), (4H, m), (5H, m); 13 C NMR (125 MHz, CDCl 3 ) δ 29.8, 30.6, 46.8, 65.1, 81.0, 95.8, 126.3, 128.3, 128.5, 140.1, 186.8; IR (CHCl 3 ) ν 3595, 3020, 2990, 2210, 1675 cm 1. HRMS (ESI) m/z calcd for C 14 H 16 O 2 Na [M+Na] + : Found: Hydroxy-2,2,6-trimethyl-4-heptyn-3-one (1d) 70% yield: Prepared from N-methoxy-N-methylpivalamide and 2-methyl- 3-butyn-2-ol. A colorless oil; 1 H NMR (500 MHz, CDCl 3 ) δ 1.20 (9H, s), 1.58 (6H, s); 13 C NMR (125 MHz, CDCl 3 ) δ 26.0, 30.7, 44.8, 65.2, 79.1, 97.0, 194.1; IR (CHCl 3 ) ν 3595, 2970, 2215, 1670 cm 1. HRMS (ESI) m/z calcd for C 10 H 16 O 2 Na [M+Na] + : Found: (1-Hydroxycyclopentyl)-1-octyn-3-one (1e) 41% yield: Prepared from N-methoxy-N-methylhexanamide and 1-ethynylcyclopentanol. A pale yellow oil; 1 H NMR (500 MHz, CDCl 3 ) δ 0.89 (3H, t, J = 7.0 Hz), (4H, m), (2H, m), (5H, m), (4H, m), 2.55 (2H, t, J = 7.4 Hz); 13 C NMR (125 MHz, CDCl 3 ) δ 13.9, 22.4, 23.56, 23.62, 31.1, 42.2, 45.4, 74.2, 82.2, 94.7, 188.2; IR (CHCl 3 ) ν 3590, 2960, 2210, 1670 cm 1. HRMS (ESI) m/z calcd for C 13 H 20 O 2 Na [M+Na] + : Found: Hydroxy-4-methyl-1-phenyl-2-pentyn-1-one (1f) 6 100% yield: Prepared from N-methoxy-N-methylbenzamide and 2-methyl-3-butyn- 2-ol. A yellow oil; 1 H NMR (500 MHz, CDCl 3 ) δ 1.67 (6H, s), 2.52 (1H, br s), 7.48 (2H, t, J = 7.7 Hz), 7.61 (1H, t, J = 7.7 Hz), 8.11 (2H, d, J = 7.7 Hz); 13 C NMR (125 MHz, CDCl 3 ) δ 30.7, 65.3, 79.9, 98.0, 128.6, 129.6, 134.2, 136.5, 177.9; IR (CHCl 3 ) ν 3595, 3015, 2215, 1645 cm 1. 6 Saimoto, H.; Yasui, M.; Ohrai, S.; Oikawa, H.; Yokoyama, K.; Shigemasa, Y. Bull. Chem. Soc. Jpn. 1999, 72, S5
6 4-Hydroxy-1-(4-methoxyphenyl)-4-methyl-2-pentyn-1-one (1g) 80% yield: Prepared from 4,N-dimethoxy-N-methylbenzamide and 2-methyl- 3-butyn-2-ol. An off-white solid, mp C; 1 H NMR (500 MHz, CDCl 3 ) δ 1.66 (6H, s), 2.30 (1H, br s), 3.89 (3H, s), 6.95 (2H, d, J = 8.8 Hz), 8.08 (2H, d, J = 8.8 Hz); 13 C NMR (125 MHz, CDCl 3 ) δ 30.8, 55.6, 65.3, 79.9, 97.0, 113.9, 129.9, 132.0, 164.6, 176.5; IR (CHCl 3 ) ν 3595, 2990, 2210, 1640 cm 1. HRMS (ESI) m/z calcd for C 13 H 14 O 3 Na [M+Na] + : Found: (1E)-6-Hydroxy-6-methyl-1-phenyl-1-hepten-4-yn-3-one (1h) 95% yield: Prepared from (2E)-N-methoxy-N-methyl-2-propenamide and 2-methyl- 3-butyn-2-ol. A pale yellow solid, mp C; 1 H NMR (500 MHz, CDCl 3 ) δ 1.65 (6H, s), 6.77 (1H, d, J = 16.5 Hz), (3H, m), (2H, m), 7.79 (1H, d, J = 16.5 Hz); 13 C NMR (125 MHz, CDCl 3 ) δ 30.8, 65.2, 79.6, 96.5, 128.1, 128.7, 129.1, 131.2, 133.9, 148.9, 178.2; IR (CHCl 3 ) ν 3595, 3435, 3015, 2990, 2220, 1630 cm 1. HRMS (ESI) m/z calcd for C 14 H 14 O 2 Na [M+Na] + : Found: Hydroxy-1-phenyl-4-hexyn-3-one (1i) 55% yield: Prepared from N-methoxy-N-methyl-3-phenylpropionamide and 1-(tert-butyldimethylsilyloxy)-2-propyne, followed by AcOH desilylation. A pale yellow oil; 1 H NMR (500 MHz, CDCl 3 ) δ 1.94 (1H, t, J = 6.3 Hz), 2.91 (2H, dt, J = 1.7, 7.7 Hz), 2.99 (2H, t, J = 7.7 Hz), 4.42 (2H, d, J = 6.3 Hz), (5H, m); 13 C NMR (125 MHz, CDCl 3 ) δ 29.7, 46.6, 50.7, 84.3, 90.2, 126.3, 128.3, 128.5, 140.0, 186.6; IR (CHCl 3 ) ν 3605, 3015, 2220, 1675 cm 1. HRMS (ESI) m/z calcd for C 12 H 12 O 2 Na [M+Na] + : Found: Pd-catalyzed coupling of thioester with alkyne to furnish 5-hydroxy-5-phenyl-3-hexyn-2-one (1c) To a solution of S-dodecyl ethanethioate (130 mg, 0.53 mmol) in DMF (0.88 ml) and Et 3 N (0.18 ml) were added PdCl 2 (dppf) CH 2 Cl 2 (21.7 mg, mmol), P(2-furyl) 3 (15.4 mg, mmol), CuI (172 mg, 0.90 mmol), and 2-phenyl-3-butyn-2-ol (155 mg, 1.1 mmol) at room temperature. After stirring for 1.5 h at 50 C, the mixture was cooled to room temperature, diluted with Et 2 O and H 2 O, and filtered through Celite. The filtrate was washed with H 2 O several times and then brine, dried over MgSO 4, and evaporated in vacuo. The residue was purified by column chromatography (silica gel, hexanes/etoac = 5:1) to give 1c (84 mg, 84%). A pale yellow oil; 1 H NMR (500 MHz, CDCl 3 ) δ 1.82 (3H, s), 2.37 (3H, s), (3H, m), (2H, m); 13 C NMR (125 MHz, CDCl 3 ) δ 32.4, 32.6, 69.8, 83.6, 93.8, 124.7, 128.2, 128.5, 143.7, 184.5; IR (CHCl 3 ) ν 3585, 3445, 3065, 2220, 1680 cm 1. HRMS (ESI) m/z calcd for C 12 H 12 O 2 Na [M+Na] + : Found: S6
7 Preparation of δ-hydroxyalkynones 3a,b 5-Hydroxy-1-phenyl-2-pentyn-1-one (3a) To a solution of 3-butyn-1-ol (0.38 ml, 5.0 mmol) in THF (15 ml) was added EtMgBr (1.0 M in THF; 11mL, 11 mmol) at 0 C under nitrogen atmosphere, and the reaction was refluxed for 1.5 h. After cooling to 78 C, a solution of benzaldehyde (523 mg, 5.0 mmol) in THF (1.5 ml) was added. The mixture was allowed to warm to room temperature, stirred overnight, quenched with sat. aq. NH 4 Cl. The organic materials were extracted with EtOAc, and the combined organic extracts were washed with brine, dried over MgSO 4, and evaporated in vacuo. The residue was purified by column chromatography (silica gel, hexanes/etoac = 1:2) to give 1-phenyl-2-pentyne-1,5-diol (776 mg, 88%). To a solution of the obtained compound (103 mg, 0.58 mmol) in CH 2 Cl 2 (12 ml) was added BaMnO 4 (1.54 g, 6.0 mmol) at room temperature under nitrogen atmosphere. The reaction was stirred for 3.5 h and filtered through Celite. The filtrate was evaporated in vacuo. The residue was purified by column chromatography (silica gel, hexanes/etoac = 1:1) to give 3a (83.1 mg, 82%). A pale yellow oil; 1 H NMR (500 MHz, CDCl 3 ) δ 2.08 (1H, br s), 2.78 (2H, t, J = 6.3 Hz), 3.91 (2H, t, J = 6.3 Hz), (2H, m), (1H, m), 8.14 (2H, dd, J = 1.2, 8.5 Hz); 13 C NMR (125 MHz, CDCl 3 ) δ 23.5, 60.3, 80.7, 93.2, 128.6, 129.6, 134.1, 136.6, 178.1; IR (CHCl 3 ) ν 3615, 3020, 2235, 2205, 1695, 1640 cm 1. HRMS (ESI) m/z calcd for C 11 H 10 O 2 Na [M+Na] + : Found: Hydroxy-1-phenyl-4-heptyn-3-one (3b) To a solution of 3-butyn-1-ol (0.421 ml, 5.6 mmol) in THF (5 ml) was added EtMgBr (1.0 M in THF; 11 ml, 11 mmol) at 0 C under nitrogen atmosphere, and the reaction was refluxed for 1.5 h. After cooling to 0 C, HMPA (2.5 ml, 14 mmol) and a solution of 3-phenylpropionaldehyde (306 mg, 2.3 mmol) in THF (5.0 ml) were added. The mixture was allowed to warm to room temperature, stirred for 4.5 h, quenched with sat. aq. NH 4 Cl. The organic materials were extracted with EtOAc, and the combined organic extracts were washed with H 2 O several times and then brine, dried over MgSO 4, and evaporated in vacuo. The residue was purified by column chromatography (silica gel, hexanes/etoac = 1:1) to give 7-phenyl-3-heptyne-1,5-diol (323 mg, 69%). To a solution of the obtained compound (116 mg, 0.57 mmol) in CH 2 Cl 2 (11 ml) was added BaMnO 4 (1.46 g, 5.7 mmol) at room temperature under nitrogen atmosphere. The reaction was stirred overnight and filtered through Celite. The filtrate was evaporated in vacuo. The residue was purified by column chromatography (silica gel, hexanes/etoac = 1:1) to give 3b (94.1 mg, 82%). A pale yellow oil; 1 H NMR (500 MHz, CDCl 3 ) δ 1.90 (1H, br s), 2.63 (2H, t, J = 6.0 Hz), (4H, m), (2H, m), (5H, m); 13 C NMR (125 MHz, CDCl 3 ) δ 23.3, 29.8, 46.8, 60.1, 81.9, 91.2, 126.3, 128.3, 128.5, 140.2, 187.0; IR (CHCl 3 ) ν 3620, 3455, 3020, 2950, 2220, 1670 cm 1. HRMS (ESI) m/z calcd for C 13 H 14 O 2 Na [M+Na] + : Found: S7
8 F:\NMR Furanone\Ysaneto NON_E3_FT.als 2-Methyl-5-phenethyl-3(2H)-furanone DATIM Wed Apr 23 20:22: EXMOD SINGL OBNUC 1H OFR MHz OBSET 0.00 KHz OBFIN Hz OBATN 511 PW usec PW usec POINT SPO SCANS 8 FREQU Hz PD sec BF 0.12 Hz IRFIN Hz IRATN 511 EXREF 7.26 ppm TMSP S8
9 F:\Saneto\Yka _ carbon.als 2-Methyl-5-phenethyl-3(2H)-furanone DATIM :31:09 EXMOD single_pulse_dec OBNUC 13C OFR MHz OBSET 7.87 KHz OBFIN 4.21 Hz PW usec POINT SPO SCANS 944 FREQU Hz PD sec EXREF ppm TMSP S9
10 F:\NMR Furanone\Yegi als 2,2-dimethyl-5-phenylethyl-3(2H)furanone DATIM :35:23 EXMOD single_pulse.ex2 OBNUC 1H OFR MHz OBSET 2.41 KHz OBFIN 6.01 Hz PW usec POINT SPO SCANS 8 FREQU Hz PD sec EXREF 7.26 ppm TMSP S10
11 F:\NMR Furanone\Yegi C.als 2,2-dimethyl-5-phenylethyl-3(2H)furanone DATIM :22:55 EXMOD single_pulse_dec OBNUC 13C OFR MHz OBSET 7.87 KHz OBFIN 4.21 Hz PW usec POINT SPO SCANS 1000 FREQU Hz PD sec EXREF ppm TMSP S11
12 F:\NMR Furanone\Yka als 2,5-dimethyl-2-phenyl-3(2H)furanone DATIM :28:42 EXMOD single_pulse.ex2 OBNUC 1H OFR MHz OBSET 2.41 KHz OBFIN 6.01 Hz PW usec POINT SPO SCANS 8 FREQU Hz PD sec EXREF 7.26 ppm TMSP S12
13 F:\NMR Furanone\Yka carbon.als 2,5-dimethyl-2-phenyl-3(2H)furanone DATIM :47:49 EXMOD single_pulse_dec OBNUC 13C OFR MHz OBSET 7.87 KHz OBFIN 4.21 Hz PW usec POINT SPO SCANS 344 FREQU Hz PD sec EXREF ppm TMSP S13
14 F:\NMR Furanone\Yka als 5-(tert-butyl)-2,2-dimethyl-3(2H)furanone DATIM :43:32 EXMOD single_pulse.ex2 OBNUC 1H OFR MHz OBSET 2.41 KHz OBFIN 6.01 Hz PW usec POINT SPO SCANS 8 FREQU Hz PD sec EXREF 7.26 ppm TMSP S14
15 F:\NMR Furanone\Yka carbon.als 5-(tert-butyl)-2,2-dimethyl-3(2H)furanone DATIM :01:31 EXMOD single_pulse_dec OBNUC 13C OFR MHz OBSET 7.87 KHz OBFIN 4.21 Hz PW usec POINT SPO SCANS 330 FREQU Hz PD sec EXREF ppm TMSP S15
16 F:\NMR Furanone\Yka als 2-pentyl-1-oxaspiro[4,4]non-2-en-4-one DATIM :24:42 EXMOD single_pulse.ex2 OBNUC 1H OFR MHz OBSET 2.41 KHz OBFIN 6.01 Hz PW usec POINT SPO SCANS 8 FREQU Hz PD sec BF 0.12 Hz EXREF 7.26 ppm TMSP S16
17 F:\NMR Furanone\Yka carbon.als 2-pentyl-1-oxaspiro[4,4]non-2-en-4-one DATIM :50:25 EXMOD single_pulse_dec OBNUC 13C OFR MHz OBSET 7.87 KHz OBFIN 4.21 Hz PW usec POINT SPO SCANS 383 FREQU Hz PD sec BF 0.12 Hz EXREF ppm TMSP S17
18 F:\NMR Furanone\Yegi-furanone (Ph,Me,Me).als 2,2-dimethyl-5-phenyl-3(2H)-furanone DATIM :50:35 EXMOD single_pulse.ex2 OBNUC 1H OFR MHz OBSET 2.41 KHz OBFIN 6.01 Hz PW usec POINT SPO SCANS 8 FREQU Hz PD sec EXREF 7.26 ppm TMSP S18
19 F:\NMR Furanone\Yegi-furanone (Ph,Me,Me)-13C.als 2,2-dimethyl-5-phenyl-3(2H)-furanone DATIM :21:14 EXMOD single_pulse_dec OBNUC 13C OFR MHz OBSET 7.87 KHz OBFIN 4.21 Hz PW usec POINT SPO SCANS 641 FREQU Hz PD sec EXREF ppm TMSP S19
20 F:\NMR Furanone\Yegi-furanone (MeOPh,Me,Me).als 5-(4-methoxyphenyl)-2,2-dimethyl-3(2H)-furanone DATIM :40:21 EXMOD single_pulse.ex2 OBNUC 1H OFR MHz OBSET 2.41 KHz OBFIN 6.01 Hz PW usec POINT SPO SCANS 8 FREQU Hz PD sec EXREF 7.26 ppm TMSP S20
21 F:\NMR Furanone\Yegi-furanone (MeOPh,Me,Me)-13C.als 5-(4-methoxyphenyl)-2,2-dimethyl-3(2H)-furanone DATIM :27:56 EXMOD single_pulse_dec OBNUC 13C OFR MHz OBSET 7.87 KHz OBFIN 4.21 Hz PW usec POINT SPO SCANS 1000 FREQU Hz PD sec EXREF ppm TMSP S21
22 F:\NMR Furanone\Yka als 2,2-Dimethyl-5-(2-phenylethenyl)-3(2H)-furanone DATIM :42:05 EXMOD single_pulse.ex2 OBNUC 1H OFR MHz OBSET 2.41 KHz OBFIN 6.01 Hz PW usec POINT SPO SCANS 8 FREQU Hz PD sec BF 0.12 Hz EXREF 7.26 ppm TMSP S22
23 F:\NMR Furanone\Yka carbon.als 2,2-Dimethyl-5-(2-phenylethenyl)-3(2H)-furanone DATIM :56:17 EXMOD single_pulse_dec OBNUC 13C OFR MHz OBSET 7.87 KHz OBFIN 4.21 Hz PW usec POINT SPO SCANS 257 FREQU Hz PD sec BF 0.12 Hz EXREF ppm TMSP S23
24 F:\NMR Furanone\Yks als 5-phenethyl-3(2H)-furanone DATIM :03:52 EXMOD single_pulse.ex2 OBNUC 1H OFR MHz OBSET 2.41 KHz OBFIN 6.01 Hz PW usec POINT SPO SCANS 8 FREQU Hz PD sec EXREF 7.26 ppm TMSP S24
25 F:\NMR Furanone\Yks carbon.als 5-phenethyl-3(2H)furanone DATIM :44:41 EXMOD single_pulse_dec OBNUC 13C OFR MHz OBSET 7.87 KHz OBFIN 4.21 Hz PW usec POINT SPO SCANS 1999 FREQU Hz PD sec EXREF ppm TMSP S25
26 F:\NMR Furanone\Yks als 6-phenyl-2,3-dihydro-4-pyranone DATIM :55:38 EXMOD single_pulse.ex2 OBNUC 1H OFR MHz OBSET 2.41 KHz OBFIN 6.01 Hz PW usec POINT SPO SCANS 8 FREQU Hz PD sec EXREF 7.26 ppm TMSP S26
27 F:\NMR Furanone\Yks carbon.als 6-phenyl-2,3-dihydro-4-pyranone DATIM :56:43 EXMOD single_pulse_dec OBNUC 13C OFR MHz OBSET 7.87 KHz OBFIN 4.21 Hz PW usec POINT SPO SCANS 1466 FREQU Hz PD sec EXREF ppm TMSP S27
28 F:\NMR Furanone\Yks als 6-phenethyl-2,3-dihydro-4-pyranone DATIM :32:24 EXMOD single_pulse.ex2 OBNUC 1H OFR MHz OBSET 2.41 KHz OBFIN 6.01 Hz PW usec POINT SPO SCANS 8 FREQU Hz PD sec BF 0.12 Hz EXREF 7.26 ppm TMSP S28
29 F:\NMR Furanone\Yks als 6-phenethyl-2,3-dihydro-4-pyranone DATIM :48:06 EXMOD single_pulse_dec OBNUC 13C OFR MHz OBSET 7.87 KHz OBFIN 4.21 Hz PW usec POINT SPO SCANS 1138 FREQU Hz PD sec BF 0.12 Hz EXREF ppm TMSP S29
30 F:\NMR Furanone\Yks als 6-hydroxy-1-phenyl-4-heptyn-3-one DATIM :09:29 EXMOD single_pulse.ex2 OBNUC 1H OFR MHz OBSET 2.41 KHz OBFIN 6.01 Hz PW usec POINT SPO SCANS 8 FREQU Hz PD sec EXREF 7.26 ppm TMSP S30
31 F:\NMR Furanone\Yks als 6-hydroxy-1-phenyl-4-heptyn-3-one DATIM :48:48 EXMOD single_pulse_dec OBNUC 13C OFR MHz OBSET 7.87 KHz OBFIN 4.21 Hz PW usec POINT SPO SCANS 2500 FREQU Hz PD sec EXREF ppm TMSP S31
32 F:\NMR Furanone\Yegi als 6-hydroxy-6-methyl-1-phenyl-4-heptyn-3-one DATIM :12:08 EXMOD single_pulse.ex2 OBNUC 1H OFR MHz OBSET 2.41 KHz OBFIN 6.01 Hz PW usec POINT SPO SCANS 8 FREQU Hz PD sec EXREF 7.26 ppm TMSP S32
33 F:\NMR Furanone\Yegi C.als 6-hydroxy-6-methyl-1-phenyl-4-heptyn-3-one DATIM :59:40 EXMOD single_pulse_dec OBNUC 13C OFR MHz OBSET 7.87 KHz OBFIN 4.21 Hz PW usec POINT SPO SCANS 1000 FREQU Hz PD sec EXREF ppm TMSP S33
34 F:\NMR Furanone\Yka als 5-hydroxy-5-phenyl-3-hexyn-2-one DATIM :08:05 EXMOD single_pulse.ex2 OBNUC 1H OFR MHz OBSET 2.41 KHz OBFIN 6.01 Hz PW usec POINT SPO SCANS 8 FREQU Hz PD sec EXREF 7.26 ppm TMSP S34
35 F:\NMR Furanone\Yka carbon.als 5-hydroxy-5-phenyl-3-hexyn-2-one DATIM :22:20 EXMOD single_pulse_dec OBNUC 13C OFR MHz OBSET 7.87 KHz OBFIN 4.21 Hz PW usec POINT SPO SCANS 254 FREQU Hz PD sec EXREF ppm TMSP S35
36 F:\NMR Furanone\Yegi-tertBu,Me,Me.als 6-hydroxy-2,2,6-trimethyl-4-heptyn-3-one DATIM :54:40 EXMOD single_pulse.ex2 OBNUC 1H OFR MHz OBSET 2.41 KHz OBFIN 6.01 Hz PW usec POINT SPO SCANS 8 FREQU Hz PD sec EXREF 7.26 ppm TMSP S36
37 F:\NMR Furanone\Yegi-tertBu,Me,Me-13C.als 6-hydroxy-2,2,6-trimethyl-4-heptyn-3-one DATIM :42:36 EXMOD single_pulse_dec OBNUC 13C OFR MHz OBSET 7.87 KHz OBFIN 4.21 Hz PW usec POINT SPO SCANS 1000 FREQU Hz PD sec EXREF ppm TMSP S37
38 F:\NMR Furanone\Yka als 1-(1-hydroxycyclopentyl)-1-octyn-3-one DATIM :03:03 EXMOD single_pulse.ex2 OBNUC 1H OFR MHz OBSET 2.41 KHz OBFIN 6.01 Hz PW usec POINT SPO SCANS 8 FREQU Hz PD sec BF 0.12 Hz EXREF 7.26 ppm TMSP S38
39 F:\NMR Furanone\Yka carbon.als 1-(1-hydroxycyclopentyl)-1-octyn-3-one DATIM :29:11 EXMOD single_pulse_dec OBNUC 13C OFR MHz OBSET 7.87 KHz OBFIN 4.21 Hz PW usec POINT SPO SCANS 469 FREQU Hz PD sec BF 0.12 Hz EXREF ppm TMSP S39
40 F:\NMR Furanone\Yka als 4-hydroxy-4-methyl-1-phenyl-2-pentyn-1-one DATIM :07:37 EXMOD single_pulse.ex2 OBNUC 1H OFR MHz OBSET 2.41 KHz OBFIN 6.01 Hz PW usec POINT SPO SCANS 8 FREQU Hz PD sec EXREF 7.26 ppm TMSP S40
41 F:\NMR Furanone\Yka carbon.als 4-hydroxy-4-methyl-1-phenyl-2-pentyn-1-one DATIM :32:44 EXMOD single_pulse_dec OBNUC 13C OFR MHz OBSET 7.87 KHz OBFIN 4.21 Hz PW usec POINT SPO SCANS 457 FREQU Hz PD sec EXREF ppm TMSP S41
42 F:\NMR Furanone\Yegi-MeOPh,Me,Me.als 4-hydroxy-1-(4-methoxyphenyl)-4-methyl-2-pentyn-1-one DATIM :56:19 EXMOD single_pulse.ex2 OBNUC 1H OFR MHz OBSET 2.41 KHz OBFIN 6.01 Hz PW usec POINT SPO SCANS 8 FREQU Hz PD sec EXREF 7.26 ppm TMSP S42
43 F:\NMR Furanone\Yegi-MeOPh,Me,Me-13C.als 4-hydroxy-1-(4-methoxyphenyl)-4-methyl-2-pentyn-1-one DATIM :35:44 EXMOD single_pulse_dec OBNUC 13C OFR MHz OBSET 7.87 KHz OBFIN 4.21 Hz PW usec POINT SPO SCANS 820 FREQU Hz PD sec EXREF ppm TMSP S43
44 F:\NMR Furanone\Yka als 1-styryl-4-hydroxy-4-methyl-2-pentyn-1-one DATIM :19:36 EXMOD single_pulse.ex2 OBNUC 1H OFR MHz OBSET 2.41 KHz OBFIN 6.01 Hz PW usec POINT SPO SCANS 8 FREQU Hz PD sec BF 0.12 Hz EXREF 7.26 ppm TMSP S44
45 F:\NMR Furanone\Yka carbon.als 1-styryl-4-hydroxy-4-methyl-2-pentyn-1-one DATIM :32:58 EXMOD single_pulse_dec OBNUC 13C OFR MHz OBSET 7.87 KHz OBFIN 4.21 Hz PW usec POINT SPO SCANS 237 FREQU Hz PD sec BF 0.12 Hz EXREF ppm TMSP S45
46 F:\NMR Furanone\Yks als 6-hydroxy-1-phenyl-4-hexyn-3-one DATIM :02:07 EXMOD single_pulse.ex2 OBNUC 1H OFR MHz OBSET 2.41 KHz OBFIN 6.01 Hz PW usec POINT SPO SCANS 8 FREQU Hz PD sec EXREF 7.26 ppm TMSP S46
47 F:\NMR Furanone\Yka carbon.als 6-hydroxy-1-phenyl-4-hexyn-3-one DATIM :51:51 EXMOD single_pulse_dec OBNUC 13C OFR MHz OBSET 7.87 KHz OBFIN 4.21 Hz PW usec POINT SPO SCANS 232 FREQU Hz PD sec EXREF ppm TMSP S47
48 F:\NMR Furanone\Yks als 5-hydroxy-1-phenyl-2-pentyn-1-one DATIM :37:48 EXMOD single_pulse.ex2 OBNUC 1H OFR MHz OBSET 2.41 KHz OBFIN 6.01 Hz PW usec POINT SPO SCANS 8 FREQU Hz PD sec EXREF 7.26 ppm TMSP S48
49 F:\NMR Furanone\Yks als 1-phenyl-5-hydroxy-2-pentyn-1-one DATIM :51:09 EXMOD single_pulse_dec OBNUC 13C OFR MHz OBSET 7.87 KHz OBFIN 4.21 Hz PW usec POINT SPO SCANS 1009 FREQU Hz PD sec BF 0.12 Hz EXREF ppm TMSP S49
50 F:\NMR Furanone\Yks pure3.als 7-hydroxy-1-phenyl-4-heptyn-3-one DATIM :37:52 EXMOD single_pulse.ex2 OBNUC 1H OFR MHz OBSET 2.41 KHz OBFIN 6.01 Hz PW usec POINT SPO SCANS 8 FREQU Hz PD sec EXREF 7.26 ppm TMSP S50
51 F:\NMR Furanone\Yks carbon.als 7-hydroxy-1-phenyl-4-heptyn-3-one DATIM :45:02 EXMOD single_pulse_dec OBNUC 13C OFR MHz OBSET 7.87 KHz OBFIN 4.21 Hz PW usec POINT SPO SCANS 1991 FREQU Hz PD sec EXREF ppm TMSP S51
Hydrosulfide adducts of organo-iridium anticancer complexes
 Hydrosulfide adducts of organo-iridium anticancer complexes Pavel Štarha, Abraha Habtemariam, Isolda Romero-Canelo n, Guy J. Clarkson, Peter J. Sadler*, Regional Centre of Advanced Technologies and Materials,
Hydrosulfide adducts of organo-iridium anticancer complexes Pavel Štarha, Abraha Habtemariam, Isolda Romero-Canelo n, Guy J. Clarkson, Peter J. Sadler*, Regional Centre of Advanced Technologies and Materials,
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFRMATIN Acid- labile mpeg- Vinyl Ether- 1,2- Dioleylglycerol Lipids with Tunable ph Sensitivity: Synthesis and Structural Effects on Hydrolysis Rates, DPE Liposome Release Performance and
SUPPLEMENTARY INFRMATIN Acid- labile mpeg- Vinyl Ether- 1,2- Dioleylglycerol Lipids with Tunable ph Sensitivity: Synthesis and Structural Effects on Hydrolysis Rates, DPE Liposome Release Performance and
Supporting Information for Achieving Accurate. Reduction Potential Predictions for. Anthraquinones in Water and Aprotic Solvents:
 Supporting Information for Achieving Accurate Reduction Potential Predictions for Anthraquinones in Water and Aprotic Solvents: Effects of Inter- and Intramolecular H-Bonding and Ion Pairing Hyungjun Kim,
Supporting Information for Achieving Accurate Reduction Potential Predictions for Anthraquinones in Water and Aprotic Solvents: Effects of Inter- and Intramolecular H-Bonding and Ion Pairing Hyungjun Kim,
Three years ( ) measurements of atmospheric concentrations of organochlorine pesticides (OCPs) at Station Nord, North East Greenland
 Three years (-) measurements of atmospheric concentrations of organochlorine pesticides (OCPs) at Station Nord, North East Greenland Rossana Bossi,* a Carsten Ambelas Skjøth,b and Henrik Skov a,c a Aarhus
Three years (-) measurements of atmospheric concentrations of organochlorine pesticides (OCPs) at Station Nord, North East Greenland Rossana Bossi,* a Carsten Ambelas Skjøth,b and Henrik Skov a,c a Aarhus
QUANTITATIVE MEASUREMENT OF FOUR MAJOR SECOIRIDOID DERIVATIVES IN OLIVE. OIL USING qnmr. PROOF OF THE ARTIFICIAL FORMATION OF ALDEHYDIC OLEUROPEIN AND
 QUANTITATIVE MEASUREMENT OF FOUR MAJOR SECOIRIDOID DERIVATIVES IN OLIVE OIL USING qnmr. PROOF OF THE ARTIFICIAL FORMATION OF ALDEHYDIC OLEUROPEIN AND LIGSTROSIDE AGLYCON ISOMERS EVANGELIA KARKOULA, ANGELIKI
QUANTITATIVE MEASUREMENT OF FOUR MAJOR SECOIRIDOID DERIVATIVES IN OLIVE OIL USING qnmr. PROOF OF THE ARTIFICIAL FORMATION OF ALDEHYDIC OLEUROPEIN AND LIGSTROSIDE AGLYCON ISOMERS EVANGELIA KARKOULA, ANGELIKI
Determination of Pesticide Residues in Tea: An AOAC Collaborative Study
 Determination of Pesticide Residues in Tea: An AOAC Collaborative Study UCT Part Numbers: RFV5CT - 5 ml centrifuge tubes ECPSACB56-6 ml SPE cartridge with 5 mg GCB and 5 mg PSA ECSS25K - Sodium sulfate,
Determination of Pesticide Residues in Tea: An AOAC Collaborative Study UCT Part Numbers: RFV5CT - 5 ml centrifuge tubes ECPSACB56-6 ml SPE cartridge with 5 mg GCB and 5 mg PSA ECSS25K - Sodium sulfate,
Appendix A. Supplementary data
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2016 Appendix A. Supplementary data Bio-oil production from eight selected green landscaping wastes
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2016 Appendix A. Supplementary data Bio-oil production from eight selected green landscaping wastes
POSS Lubricants and Blends
 PSS Lubricants and Blends usty L. Blanski, Shawn H. Phillips, Joe Lichtenhan, Joe Schwab, Andre Lee, Justin Leland, and Patrick uth AFL/PSM 10 E. Saturn Blvd. Edwards AFB CA 93524 661-525-5391 Edwards
PSS Lubricants and Blends usty L. Blanski, Shawn H. Phillips, Joe Lichtenhan, Joe Schwab, Andre Lee, Justin Leland, and Patrick uth AFL/PSM 10 E. Saturn Blvd. Edwards AFB CA 93524 661-525-5391 Edwards
Bunsen burner and attached tubing, striker, evaporating dish, wire gauze pad, tongs.
 LABORATORY PROCEDURES OBJECTIVES: To observe proper safety techniques with all laboratory equipment. To use laboratory apparatus skillfully and efficiently. Before you actually complete an experiment,
LABORATORY PROCEDURES OBJECTIVES: To observe proper safety techniques with all laboratory equipment. To use laboratory apparatus skillfully and efficiently. Before you actually complete an experiment,
Annex 1. New psychoactive substances first notified to the Early Warning System in 2015 under the terms of Council Decision 2005/387/JHA
 Annex 1. New psychoactive substances first notified to the Early Warning System in under the terms of Council Decision 2005/387/JHA 1. 5F-MDMB-PINACA (methyl-[2-(1-(5-fluoropentyl)-1h-indazole-3-carboxamido)-
Annex 1. New psychoactive substances first notified to the Early Warning System in under the terms of Council Decision 2005/387/JHA 1. 5F-MDMB-PINACA (methyl-[2-(1-(5-fluoropentyl)-1h-indazole-3-carboxamido)-
CHAPTER 8 SOLUTIONS TO EXERCISES
 CHAPTER 8 SOLUTIONS TO EXERCISES Solution 8.2. The walk starts with the anomeric proton (peak h), which can be unambiguously assigned because it is downfield of all the other peaks, and it is the only
CHAPTER 8 SOLUTIONS TO EXERCISES Solution 8.2. The walk starts with the anomeric proton (peak h), which can be unambiguously assigned because it is downfield of all the other peaks, and it is the only
Vivaspin 500, 2, 6 and 20
 GE Healthcare Instructions 28-9331-02 AB Vivaspin 500, 2, 6 and 20 Intended use This product is intended for research use only, and shall not be used in any clinical or in vitro procedures for diagnostic
GE Healthcare Instructions 28-9331-02 AB Vivaspin 500, 2, 6 and 20 Intended use This product is intended for research use only, and shall not be used in any clinical or in vitro procedures for diagnostic
Origin and Extraction of Andean Salars
 Think Simulation! Adventures in Electrolytes Origin and Extraction of Andean Salars Lithium and Boron Recovery from Salt brines and Salt flats in Chile and Bolivia http://tunari.tripod.com/landscapes.html
Think Simulation! Adventures in Electrolytes Origin and Extraction of Andean Salars Lithium and Boron Recovery from Salt brines and Salt flats in Chile and Bolivia http://tunari.tripod.com/landscapes.html
Chemical Resistance PPA 571
 Chemical Resistance PPA 571 - Prolonged contact refers to the process where the coating material is immersed in the chemical permanently, or intermittently for long periods of time. - Occasional short
Chemical Resistance PPA 571 - Prolonged contact refers to the process where the coating material is immersed in the chemical permanently, or intermittently for long periods of time. - Occasional short
TELOS Silica-based Solid Phase Extraction Columns From Kinesis
 TELOS Silica-based Solid Phase Extraction Columns From Kinesis Overview TELOS Non-polar (Reversed Phase) SPE Sorbents C18 C18(EC) C18 AQ(EC) C8(EC) C2(EC) TELOS Ion Exchange Sorbents SCX SAX NH2 TELOS
TELOS Silica-based Solid Phase Extraction Columns From Kinesis Overview TELOS Non-polar (Reversed Phase) SPE Sorbents C18 C18(EC) C18 AQ(EC) C8(EC) C2(EC) TELOS Ion Exchange Sorbents SCX SAX NH2 TELOS
Nexus Standard Glove Box System
 NOW CONFORMS TO UL AND CSA STANDARDS FOR LABORATORY EQUIPMENT Nexus Standard Glove Box System Vacuum Atmospheres Company Vacuum Atmospheres Company 4652 W. Rosecrans Ave. East Coast Office P.O. Box 1043
NOW CONFORMS TO UL AND CSA STANDARDS FOR LABORATORY EQUIPMENT Nexus Standard Glove Box System Vacuum Atmospheres Company Vacuum Atmospheres Company 4652 W. Rosecrans Ave. East Coast Office P.O. Box 1043
Titan3 Syringe Filters. Sample Preparation 1-036
 Sample Preparation Titan3 Syringe Filters Low extractable membranes and housing HPLC performance tested Color coding for easy selection of the correct membrane and pore size Enhanced Luer Lock inlet which
Sample Preparation Titan3 Syringe Filters Low extractable membranes and housing HPLC performance tested Color coding for easy selection of the correct membrane and pore size Enhanced Luer Lock inlet which
Innovative New Products
 Innovative New Products NEW Screw Top Micro Tubes Product Family Categories: Sterile Assemblies (Tube with Green Grip ) Only (Grip, Opaque, Translucent ) Tube Only (Conical Bottom & Self-Standing) High
Innovative New Products NEW Screw Top Micro Tubes Product Family Categories: Sterile Assemblies (Tube with Green Grip ) Only (Grip, Opaque, Translucent ) Tube Only (Conical Bottom & Self-Standing) High
******* 4 TH SECTION (PART I & II) *******
 ******* 4 TH SECTION (PART I & II) ******* Methods of increasing selectivity of is very unselective because it can form stable complexes with many divalent, trivalent and tetravalent cations. Many methods
******* 4 TH SECTION (PART I & II) ******* Methods of increasing selectivity of is very unselective because it can form stable complexes with many divalent, trivalent and tetravalent cations. Many methods
!"#$%&'()*+,-./012 ()*+,-. /0*+,123.
 !"#$%&'()*,.//0"34 (C) C a Ts(C ) 3Ts a!"#$%&'()*,./0 ()*,. /0*,3. 567(8(9%&'():;
!"#$%&'()*,.//0"34 (C) C a Ts(C ) 3Ts a!"#$%&'()*,./0 ()*,. /0*,3. 567(8(9%&'():;
Texas Commission on Environmental Quality
 Method 40 CFR Part 50 Appendix L AB ID Method ID Fine particulates
Method 40 CFR Part 50 Appendix L AB ID Method ID Fine particulates
P R O D U C T C A T A L O G U E 2013
 PRODUCT CATALOGUE 2013 There is a place in Poland, with lush nature and magnificent views. Nowhere else is the air and water as crystal clear as there. The Pieniny count amongst the purest mountain areas
PRODUCT CATALOGUE 2013 There is a place in Poland, with lush nature and magnificent views. Nowhere else is the air and water as crystal clear as there. The Pieniny count amongst the purest mountain areas
CW1308 Internal Telephone Cable HFFR or PVC Sheath
 Applications Internal Telephone Cable for interconnection of telephone systems and other communications equipment. Cross Section Drawing Insulation PET Tape Sheath Rip Cord Design Unit Insulation Twinning
Applications Internal Telephone Cable for interconnection of telephone systems and other communications equipment. Cross Section Drawing Insulation PET Tape Sheath Rip Cord Design Unit Insulation Twinning
Old-Style Aging Cell 316 Stainless Steel mL Capacity. Part No Instruction Manual. Updated 8/21/2014 Ver. 1.7
 Old-Style Aging Cell 316 Stainless Steel - 500-mL Capacity Part No. 175-70 Instruction Manual Updated 8/21/2014 Ver. 1.7 OFI Testing Equipment, Inc. 11302 Steeplecrest Dr. Houston, Texas 77065 U.S.A. Tele:
Old-Style Aging Cell 316 Stainless Steel - 500-mL Capacity Part No. 175-70 Instruction Manual Updated 8/21/2014 Ver. 1.7 OFI Testing Equipment, Inc. 11302 Steeplecrest Dr. Houston, Texas 77065 U.S.A. Tele:
PACE APTITUDE & TALENT HUNT
 PACE APTITUDE & TALENT HUNT MUMBAI / NASHIK / PUNE / AKOLA / GOA / DELHI / LUCKNOW / KOLKATA ANSWERS KEY PAPER CODE-31 PATH TEST STUDENTS CURRENTLY STUDYING IN CLASS VI DATE: 04/09/2016 Q. No. Answer Q.
PACE APTITUDE & TALENT HUNT MUMBAI / NASHIK / PUNE / AKOLA / GOA / DELHI / LUCKNOW / KOLKATA ANSWERS KEY PAPER CODE-31 PATH TEST STUDENTS CURRENTLY STUDYING IN CLASS VI DATE: 04/09/2016 Q. No. Answer Q.
Laboratories & Consulting Group
 Final Report Efficacy of Hypobromous Acid as a Hide Intervention Performed July 18-20, 2011 Submitted to Mike Harvey Technical Operations Mgr. Enviro Tech 500 Winmoore Way Modesto, CA 95358 209-232-2211
Final Report Efficacy of Hypobromous Acid as a Hide Intervention Performed July 18-20, 2011 Submitted to Mike Harvey Technical Operations Mgr. Enviro Tech 500 Winmoore Way Modesto, CA 95358 209-232-2211
Disposable Protective Apparel
 Disposable Protective Apparel BRC represents quality suppliers of professionally manufactured disposable safety and healthcare products from China and Taiwan. All of our N95 Respirators meet the European
Disposable Protective Apparel BRC represents quality suppliers of professionally manufactured disposable safety and healthcare products from China and Taiwan. All of our N95 Respirators meet the European
B 270 Superwite D Requirements deviating from these specifications must be defined in writing in a customer agreement.
 B 270 uperwite B 270 uperwite D 0092 B 270 uperwite is a clear high transmission crown glass (modified soda-lime glass) available in form of sheets, optical rods, profiled rods, strips and chain moulded
B 270 uperwite B 270 uperwite D 0092 B 270 uperwite is a clear high transmission crown glass (modified soda-lime glass) available in form of sheets, optical rods, profiled rods, strips and chain moulded
Supporting Information
 Supporting Information Synthesis Of Novel Derivatives Of Thiophene- Carbazole, Their Electronic Properties And Computational Studies Damit E. F., 1 Nordin N., 1 Ariffin A.*,1 and Sulaiman K. 2 1 Chemistry
Supporting Information Synthesis Of Novel Derivatives Of Thiophene- Carbazole, Their Electronic Properties And Computational Studies Damit E. F., 1 Nordin N., 1 Ariffin A.*,1 and Sulaiman K. 2 1 Chemistry
Gas Chromatographic Presumptive Test for Coliform Bacteria in Water
 AmPID MICROBIOLOGY, Oct. 1975, P. 584-588 Copyright X) 1975 American Society for Microbiology Vol. 30, No. 4 Printed in U.SA. Gas Chromatographic Presumptive Test for Coliform Bacteria in Water JUDITH
AmPID MICROBIOLOGY, Oct. 1975, P. 584-588 Copyright X) 1975 American Society for Microbiology Vol. 30, No. 4 Printed in U.SA. Gas Chromatographic Presumptive Test for Coliform Bacteria in Water JUDITH
Investigation of the effect of antibiotics on bacterial growth. Introduction. Apparatus. Diagram of Apparatus
 Investigation of the effect of antibiotics on bacterial growth Introduction Antimicrobials are agents that are able to kill bacteria or halt their growth. They are widely used in medicine to treat bacterial
Investigation of the effect of antibiotics on bacterial growth Introduction Antimicrobials are agents that are able to kill bacteria or halt their growth. They are widely used in medicine to treat bacterial
Temperature affects the silicate morphology in a diatom
 Supplementary Information: Temperature affects the silicate morphology in a diatom N. Javaheri 1, R. Dries 1&2, A. Burson 3, L.J. Stal 3&4, P.M.A. Sloot 1,5,6, J.A. Kaandorp 1* 1- Computational Science,
Supplementary Information: Temperature affects the silicate morphology in a diatom N. Javaheri 1, R. Dries 1&2, A. Burson 3, L.J. Stal 3&4, P.M.A. Sloot 1,5,6, J.A. Kaandorp 1* 1- Computational Science,
Silica-based Solid Phase Extraction Columns
 Silica-based Solid Phase Extraction Columns kinesis-group.com Contents TELOS Non-polar (Reversed Phase) SPE Sorbents Page 4 Page 5 Page 5 Page 6 Page 6 C18 C18(EC) C18 AQ(EC) C8(EC) C2(EC) TELOS Ion Exchange
Silica-based Solid Phase Extraction Columns kinesis-group.com Contents TELOS Non-polar (Reversed Phase) SPE Sorbents Page 4 Page 5 Page 5 Page 6 Page 6 C18 C18(EC) C18 AQ(EC) C8(EC) C2(EC) TELOS Ion Exchange
Bacterial Quality of Crystalline Rock and Glacial Aquifers in New England
 Bacterial Quality of Crystalline Rock and Glacial Aquifers in New England By Sarah Flanagan and Charles Culbertson, U.S. Geological Survey 2012 New Hampshire Water and Watershed Conference Plymouth State
Bacterial Quality of Crystalline Rock and Glacial Aquifers in New England By Sarah Flanagan and Charles Culbertson, U.S. Geological Survey 2012 New Hampshire Water and Watershed Conference Plymouth State
Silica-based Solid Phase Extraction Columns
 Silica-based Solid Phase Extraction Columns Kinesis Ltd kinesis.co.uk Kinesis Inc kinesis-usa.com Kinesis GmbH (formerly Abimed) kinesisgmbh.de Kinesis Australia Pty Ltd kinesis-australia.com.au Contents
Silica-based Solid Phase Extraction Columns Kinesis Ltd kinesis.co.uk Kinesis Inc kinesis-usa.com Kinesis GmbH (formerly Abimed) kinesisgmbh.de Kinesis Australia Pty Ltd kinesis-australia.com.au Contents
Making Solar Cells 1 from Household Materials (mostly :)
 Making Solar Cells 1 from Household Materials (mostly :) Equipment and Materials List Safety Glasses (20+ pairs for students and instructors) Nitrile Gloves (Supreno, 15 pairs of small or medium for students,
Making Solar Cells 1 from Household Materials (mostly :) Equipment and Materials List Safety Glasses (20+ pairs for students and instructors) Nitrile Gloves (Supreno, 15 pairs of small or medium for students,
- Make sure the anaerobic tent will be available on the days you plan to use it. - Make sure the autoclave/incubator will be available.
 LytB Procedures - Make sure the anaerobic tent will be available on the days you plan to use it. - Make sure the autoclave/incubator will be available. - For the transformation of the host cells (E. coli
LytB Procedures - Make sure the anaerobic tent will be available on the days you plan to use it. - Make sure the autoclave/incubator will be available. - For the transformation of the host cells (E. coli
WÄRTSILÄ EXHAUST GAS CLEANING SYSTEM. Stian Aakre General Manager R&D. 1 Wärtsilä
 WÄRTSILÄ EXHAUST GAS CLEANING SYSTEM Stian Aakre General Manager R&D 1 Wärtsilä BPO Environmental Seminar 5 th Mach 2014 Wärtsilä scrubber system technology options Types and amounts of waste 2 Wärtsilä
WÄRTSILÄ EXHAUST GAS CLEANING SYSTEM Stian Aakre General Manager R&D 1 Wärtsilä BPO Environmental Seminar 5 th Mach 2014 Wärtsilä scrubber system technology options Types and amounts of waste 2 Wärtsilä
Introduction TEFLON PFA Filtration
 Introduction TEFLON PFA Filtration The following pages are devoted to in-line filtration devices. Designs follow the popular 47 and 50 holders manufactured the past 23 years by Savillex Corp. Typical applications
Introduction TEFLON PFA Filtration The following pages are devoted to in-line filtration devices. Designs follow the popular 47 and 50 holders manufactured the past 23 years by Savillex Corp. Typical applications
Minisart Syringe Filters
 Minisart Syringe Filters Removal of Particles and Microorganisms from Liquids and Gases Sample Preparation HPLC UHPLC Analytics Elimination of particles from your samples prior to HPLC or other chromatographic
Minisart Syringe Filters Removal of Particles and Microorganisms from Liquids and Gases Sample Preparation HPLC UHPLC Analytics Elimination of particles from your samples prior to HPLC or other chromatographic
Int. Res J Pharm. App Sci., 2013; 3(5): ISSN:
 International Research Journal of Pharmaceutical and Applied Sciences (IRJPAS) Available online at www.irjpas.com Int. Res J Pharm. App Sci., 2013; 3(5):168-174 Research Article DEVELOPMENT AND VALIDATION
International Research Journal of Pharmaceutical and Applied Sciences (IRJPAS) Available online at www.irjpas.com Int. Res J Pharm. App Sci., 2013; 3(5):168-174 Research Article DEVELOPMENT AND VALIDATION
SQUARE JARS Jar 250gm / 200ml Amber Square Pallet (3648 per pallet) 57mm neck Jar 250gm / 200ml Amber Square Carton (336 per carton) 57mm neck
 Mossop s Honey Date: 3 rd May 2016 761 State Highway 29 Tauriko RD1, Tauranga 3171 Attention: Wendy Mossop Dear Wendy, This is to confirm that products for Mossop s Honey as listed are manufactured as
Mossop s Honey Date: 3 rd May 2016 761 State Highway 29 Tauriko RD1, Tauranga 3171 Attention: Wendy Mossop Dear Wendy, This is to confirm that products for Mossop s Honey as listed are manufactured as
Pb 2+ Complete exchange 2 days. Zn 2+ Partial node exchange. Cd ncl2. 2 Cd 1.5(H3O)3[(Cd4O)3(HETT)8] 1 Zn Zn3[(Zn0.7Mn3.
![Pb 2+ Complete exchange 2 days. Zn 2+ Partial node exchange. Cd ncl2. 2 Cd 1.5(H3O)3[(Cd4O)3(HETT)8] 1 Zn Zn3[(Zn0.7Mn3. Pb 2+ Complete exchange 2 days. Zn 2+ Partial node exchange. Cd ncl2. 2 Cd 1.5(H3O)3[(Cd4O)3(HETT)8] 1 Zn Zn3[(Zn0.7Mn3.](/thumbs/94/121762313.jpg) Linker Original Linker Name L1 BTT = 1,3,5- benzenetristetrazolate L2 (HETT = 5,5,10,10,15,15 - Hexaethayltruxene- 2,7,12- MOF Original MOF Name Parent Metal Exchanged Metal Additional Notes Ref. 1 Mn
Linker Original Linker Name L1 BTT = 1,3,5- benzenetristetrazolate L2 (HETT = 5,5,10,10,15,15 - Hexaethayltruxene- 2,7,12- MOF Original MOF Name Parent Metal Exchanged Metal Additional Notes Ref. 1 Mn
JULIAN DEAN, PETER IVANOV, SEAN COLLINS AND MARIA GARCIA MIRANDA
 NPL REPORT IR 32 Environmental Radioactivity Proficiency Test Exercise 2013 JULIAN DEAN, PETER IVANOV, SEAN COLLINS AND MARIA GARCIA MIRANDA JULY 2014 Environmental Radioactivity Proficiency Test Exercise
NPL REPORT IR 32 Environmental Radioactivity Proficiency Test Exercise 2013 JULIAN DEAN, PETER IVANOV, SEAN COLLINS AND MARIA GARCIA MIRANDA JULY 2014 Environmental Radioactivity Proficiency Test Exercise
Core practical 13: Isolate an individual species from a mixed culture of bacteria using streak plating
 Core practical 13 Teacher sheet Core practical 13: Isolate an individual species from a mixed culture of Objectives To successfully use streak plating techniques to isolate a single species of bacteria
Core practical 13 Teacher sheet Core practical 13: Isolate an individual species from a mixed culture of Objectives To successfully use streak plating techniques to isolate a single species of bacteria
Operating Instructions
 Operating Instructions High Throughput Dialysis MODEL HTD96b www.htdialysis.com HTD96b : Operating Instructions Components Universal Base Unit Teflon block assembly: 9 Teflon bars, labeled sequentially
Operating Instructions High Throughput Dialysis MODEL HTD96b www.htdialysis.com HTD96b : Operating Instructions Components Universal Base Unit Teflon block assembly: 9 Teflon bars, labeled sequentially
OREGON Environmental Laboratory Accreditation Program ORELAP Fields of Accreditation BSK Associates Vancouver
 MATRIX Reference Code Analyte Code Description Drinking EPA 300.0 2.1 10053200 Methods for the Determination of Inorganic Substances in Environmental Samples 1575 Chloride 1730 Fluoride 1810 Nitrate as
MATRIX Reference Code Analyte Code Description Drinking EPA 300.0 2.1 10053200 Methods for the Determination of Inorganic Substances in Environmental Samples 1575 Chloride 1730 Fluoride 1810 Nitrate as
INTRODUCTION. Materials. Materials MEASURING MATTER. Contents. NOTE Delta Education Customer Service can be reached at
 MEASURING MATTER Materials Materials Contents Introduction... 37 Kit Inventory List... 38 Materials Supplied by the Teacher... 40 Preparing a New Kit... 42 Preparing the Kit for Your Classroom... 44 Care,
MEASURING MATTER Materials Materials Contents Introduction... 37 Kit Inventory List... 38 Materials Supplied by the Teacher... 40 Preparing a New Kit... 42 Preparing the Kit for Your Classroom... 44 Care,
Engine Thrust Reverser Emissions at Zurich Airport
 Engine Emissions at Zurich Airport - 2 - Emissions Content 1 Introduction...3 2 Airport...3 2.1 Airport Layout and Conditions...3 2.2 Regulation...3 3 Operational Data...4 4 Fuel Flow and Emission Factors...5
Engine Emissions at Zurich Airport - 2 - Emissions Content 1 Introduction...3 2 Airport...3 2.1 Airport Layout and Conditions...3 2.2 Regulation...3 3 Operational Data...4 4 Fuel Flow and Emission Factors...5
Category Opening Hours Place. September 21 (Wed) - 23 (Fri), Grand Ballroom Left-Lobby, Convention Hall 1F, Coex Registration Desk (Post Congress)
 General Information General Information Onsite Opening Hours Category Opening Hours Place Registration Desk September 21 (Wed) - 23 (Fri), Grand Ballroom Left-Lobby, 07:00-18:00 Convention Hall 1F, Coex
General Information General Information Onsite Opening Hours Category Opening Hours Place Registration Desk September 21 (Wed) - 23 (Fri), Grand Ballroom Left-Lobby, 07:00-18:00 Convention Hall 1F, Coex
9 LABORATORY ACCESSORY
 9 NORMAG - LABORATORY GLASSWARE L 309 e.1 DRYING TUBES They serve for drying of gas with suitable desiccants. To avoid sticking of the packing during the process, not shape resistant desiccants (e.g. phosphorus
9 NORMAG - LABORATORY GLASSWARE L 309 e.1 DRYING TUBES They serve for drying of gas with suitable desiccants. To avoid sticking of the packing during the process, not shape resistant desiccants (e.g. phosphorus
Toyostove Error Code EE 2 (EE 6)
 Toyostove Error Code EE 2 (EE 6) WARNING Rural Energy Enterprises, Inc. does not accept liability for the improper use of this information. Installation, service, and maintenance of heating equipment should
Toyostove Error Code EE 2 (EE 6) WARNING Rural Energy Enterprises, Inc. does not accept liability for the improper use of this information. Installation, service, and maintenance of heating equipment should
1) Date, 2) Partner, 3) Title, 4) Purpose, 5) Materials, 6) Safety, 7) Diagram, 8) Procedures, and 9) Data Table.
 LAB: BUNSEN BURNER Students: Please read the following information given below, and then come to class on your lab day with the following already prepared in your notebooks: 1) Date, 2) Partner, 3) Title,
LAB: BUNSEN BURNER Students: Please read the following information given below, and then come to class on your lab day with the following already prepared in your notebooks: 1) Date, 2) Partner, 3) Title,
High Speed Centrifuge. LHS-A, B Series
 High Speed Centrifuge LHS-A, B Series info@labtron.com www.labtron.com High Speed Centrifuge LHS-A10 High Speed Centrifuge LHS-A10 is a microprocessor controlled light weight table top type system with
High Speed Centrifuge LHS-A, B Series info@labtron.com www.labtron.com High Speed Centrifuge LHS-A10 High Speed Centrifuge LHS-A10 is a microprocessor controlled light weight table top type system with
Water Filtration & Purification System
 INSTALLATION AND OPERATION INSTR UCTIONS FOR Series 200 High Flow Water Filtration & Purification System Table of Contents A. General Information B. Specifications C. Component Description D. Installation
INSTALLATION AND OPERATION INSTR UCTIONS FOR Series 200 High Flow Water Filtration & Purification System Table of Contents A. General Information B. Specifications C. Component Description D. Installation
Ceramic Probe Replacement Instructions Accessory G
 Ceramic Probe Replacement Instructions Accessory G6600-60035 Purpose The Stainless Burner has two ceramic combustion tubes, which require periodic replacement: an inner and an outer tube. Scope This procedure
Ceramic Probe Replacement Instructions Accessory G6600-60035 Purpose The Stainless Burner has two ceramic combustion tubes, which require periodic replacement: an inner and an outer tube. Scope This procedure
Installing the Agilent 6890 Oven Insert for Fast Chromatography
 Installing the Agilent 6890 Oven Insert for Fast Chromatography Kit G2646-60500 The 6890 Oven Insert for Fast Chromatography reduces the oven volume so that the column and sample heat more quickly, yielding
Installing the Agilent 6890 Oven Insert for Fast Chromatography Kit G2646-60500 The 6890 Oven Insert for Fast Chromatography reduces the oven volume so that the column and sample heat more quickly, yielding
Development of Stove running on low ethanol concentration
 1 Development of Stove running on low ethanol concentration Anil K. Rajvanshi, S. M. Patil and B. Mendoca Nimbkar Agricultural Research Institute (NARI), P.O. Box 44, PHALTAN-415523, Maharashtra, India
1 Development of Stove running on low ethanol concentration Anil K. Rajvanshi, S. M. Patil and B. Mendoca Nimbkar Agricultural Research Institute (NARI), P.O. Box 44, PHALTAN-415523, Maharashtra, India
MODEL Task Team. NEMURO NEMURO.FISH Dynamic link Coupled with population dynamics model PEST Regional comparison 3D-NEMURO
 MODEL Task Team NEMURO NEMURO.FISH Dynamic link Coupled with population dynamics model PEST Regional comparison E-NEMURO 3D-NEMURO ECOPATH/ECOSIM+NEMURO Ecological Modelling special volume Future perspectives
MODEL Task Team NEMURO NEMURO.FISH Dynamic link Coupled with population dynamics model PEST Regional comparison E-NEMURO 3D-NEMURO ECOPATH/ECOSIM+NEMURO Ecological Modelling special volume Future perspectives
Penberthy Series LL, LM, LH, GL and GH Jet Pumps for Pumping Liquids
 Practical, simple and cost-effective alternatives for process industries to pump a range of liquids Features Simple design with no moving parts to wear out. No lubrication required. Virtually maintenance-free.
Practical, simple and cost-effective alternatives for process industries to pump a range of liquids Features Simple design with no moving parts to wear out. No lubrication required. Virtually maintenance-free.
3M Purifi cation Life Sciences Process Technologies Product Brochure Zeta Plus Encapsulated Filter System Innovative Filtration. Fast, Easy and Clean.
 3M Purification Life Sciences Process Technologies Product Brochure Zeta Plus Encapsulated Filter System Innovative Filtration. Fast, Easy and Clean. Zeta Plus encapsulated standard capsule offering with
3M Purification Life Sciences Process Technologies Product Brochure Zeta Plus Encapsulated Filter System Innovative Filtration. Fast, Easy and Clean. Zeta Plus encapsulated standard capsule offering with
Thomas Scientific Swedesboro, NJ U.S.A.
 Thomas Scientific Swedesboro, NJ 08085-0099 U.S.A. Wiley Laboratory Mill, Model 4 3375-E10 (115 V, 50/60 HZ) USE AND CARE OF CATALOG NUMBER: 3375-E10 Wiley Laboratory Mill, Model 4 (115 V, 50/60 HZ) UNPACKING
Thomas Scientific Swedesboro, NJ 08085-0099 U.S.A. Wiley Laboratory Mill, Model 4 3375-E10 (115 V, 50/60 HZ) USE AND CARE OF CATALOG NUMBER: 3375-E10 Wiley Laboratory Mill, Model 4 (115 V, 50/60 HZ) UNPACKING
PM 2.5 source apportionment in the Po Valley: a 4 city study in Emilia-Romagna region
 PM 2.5 source apportionment in the Po Valley: a 4 city study in Emilia-Romagna region F. Scotto 1, I. Ricciardelli 1, D. Bacco 1, A. Vagheggini 2, A. Trentini 1, S. Ferrari 1, V. Poluzzi 1, C. Maccone
PM 2.5 source apportionment in the Po Valley: a 4 city study in Emilia-Romagna region F. Scotto 1, I. Ricciardelli 1, D. Bacco 1, A. Vagheggini 2, A. Trentini 1, S. Ferrari 1, V. Poluzzi 1, C. Maccone
Geochemistry of Surface Water and Groundwater on the Campus of The Ohio State University. Senior Thesis
 Geochemistry of Surface Water and Groundwater on the Campus of The Ohio State University Senior Thesis Submitted in partial fulfillment of the requirements for the Bachelor of Science Degree, with Research
Geochemistry of Surface Water and Groundwater on the Campus of The Ohio State University Senior Thesis Submitted in partial fulfillment of the requirements for the Bachelor of Science Degree, with Research
Caribbean Coastal Pollution Project (CCPP)
 Caribbean Coastal Pollution Project (CCPP) Tracking the Sources of Coastal Zone Contamination in the Maya Riviera Region of Mexico Report on Research Project December 11, 2009 United Nations University
Caribbean Coastal Pollution Project (CCPP) Tracking the Sources of Coastal Zone Contamination in the Maya Riviera Region of Mexico Report on Research Project December 11, 2009 United Nations University
p u m p s Technology. Innovation. Commitment. S T A I N L E S S S T E E L S U B M E R S I B L E P U M P S E n h a n c e y o u r l i f e
 p u m p s Technology. Commitment. Innovation. S T A I N L E S S S T E E L S U B M E R S I B L E P U M P S E n h a n c e y o u r l i f e Company Profile - 6 Description & Pumped Liquids 7 The Inside Picture
p u m p s Technology. Commitment. Innovation. S T A I N L E S S S T E E L S U B M E R S I B L E P U M P S E n h a n c e y o u r l i f e Company Profile - 6 Description & Pumped Liquids 7 The Inside Picture
Annual Weather Book RECORDED BY NW RESEARCH & OUTREACH CTR. By: Michael Leiseth
 Annual Weather Book RECORDED BY NW RESEARCH & OUTREACH CTR. By: Michael Leiseth Table I II 1 2 3 4 5 6 7 8 9, 10 11, 12 13, 14 15, 16 17 18 An average year in Crookston, MN. Seasonal extremes in Crookston,
Annual Weather Book RECORDED BY NW RESEARCH & OUTREACH CTR. By: Michael Leiseth Table I II 1 2 3 4 5 6 7 8 9, 10 11, 12 13, 14 15, 16 17 18 An average year in Crookston, MN. Seasonal extremes in Crookston,
Lab Equipment. Perry High School. Your lab equipment should:
 Lab Equipment Perry High School Your lab equipment should: 1. Be CLEAN before using it. 2. Be CHECKED (if glassware) for cracks, broken edges, and stars discard anything damaged. 3. Be washed, dried, and
Lab Equipment Perry High School Your lab equipment should: 1. Be CLEAN before using it. 2. Be CHECKED (if glassware) for cracks, broken edges, and stars discard anything damaged. 3. Be washed, dried, and
Withdrawal of Pesticide Petitions for Establishment or Modification of Pesticide Chemical Tolerances or Tolerance Exemptions
 This document is scheduled to be published in the Federal Register on 01/19/2016 and available online at http://federalregister.gov/a/2016-00431, and on FDsys.gov ENVIRONMENTAL PROTECTION AGENCY 40 CFR
This document is scheduled to be published in the Federal Register on 01/19/2016 and available online at http://federalregister.gov/a/2016-00431, and on FDsys.gov ENVIRONMENTAL PROTECTION AGENCY 40 CFR
SIMULTANEOUS RP HPLC METHOD DEVELOPMENT AND VALIDATION OF LEVOFLOXACIN AND ORNIDAZOLE IN COMBINED PHARMACEUTICAL DOSAGE FORMS
 Int. J. Chem. Sci.: 8(4), 2010, 2145-2152 SIMULTANEOUS RP HPLC METHOD DEVELOPMENT AND VALIDATION OF LEVOFLOXACIN AND ORNIDAZOLE IN COMBINED PHARMACEUTICAL DOSAGE FORMS CH. NARASIMHA RAJU BH *, K. V. RAMANA,
Int. J. Chem. Sci.: 8(4), 2010, 2145-2152 SIMULTANEOUS RP HPLC METHOD DEVELOPMENT AND VALIDATION OF LEVOFLOXACIN AND ORNIDAZOLE IN COMBINED PHARMACEUTICAL DOSAGE FORMS CH. NARASIMHA RAJU BH *, K. V. RAMANA,
UNITED STATES MARINE CORPS FIELD MEDICAL TRAINING BATTALION Camp Lejeune, NC
 UNITED STATES MARINE CORPS FIELD MEDICAL TRAINING BATTALION Camp Lejeune, NC 28542-0042 FMST 201 Perform Water Purification for Individual Use TERMINAL LEARNING OBJECTIVES 1. Given water and hygiene items,
UNITED STATES MARINE CORPS FIELD MEDICAL TRAINING BATTALION Camp Lejeune, NC 28542-0042 FMST 201 Perform Water Purification for Individual Use TERMINAL LEARNING OBJECTIVES 1. Given water and hygiene items,
Wetlands influencing river biogeochemistry: the case study of the Zambezi and the Kafue Rivers
 AFRIVAL Project (African River Basins) http://ees.kuleuven.be/project/afrival/ EGU General Assembly 2014, Vienna Wetlands influencing river biogeochemistry: the case study of the Zambezi and the Kafue
AFRIVAL Project (African River Basins) http://ees.kuleuven.be/project/afrival/ EGU General Assembly 2014, Vienna Wetlands influencing river biogeochemistry: the case study of the Zambezi and the Kafue
MethaShip Methanol as fuel for engines in passenger shipping
 Evaluation of Methanol MethaShip Methanol as fuel for engines in passenger shipping Methanol Technical Workshop Copenhagen, 20. March 2018 MEYER WERFT at a glance Family owned in 6th generation 3200 employees
Evaluation of Methanol MethaShip Methanol as fuel for engines in passenger shipping Methanol Technical Workshop Copenhagen, 20. March 2018 MEYER WERFT at a glance Family owned in 6th generation 3200 employees
Silicon Wafers. Specifications. Front side Finish: Polished Back side Finish: Etched Primary Flat: Semi
 Silicon Wafers 2 inch p-type test wafers 2 inch n-type wafers Material: CZ Silicon Front side Finish: Polished Back side Finish: Etched Primary Flat: Semi Resistivity: min:1 to max:10 Ohm/cm N-Type Item#
Silicon Wafers 2 inch p-type test wafers 2 inch n-type wafers Material: CZ Silicon Front side Finish: Polished Back side Finish: Etched Primary Flat: Semi Resistivity: min:1 to max:10 Ohm/cm N-Type Item#
INTRODUCING A NEW KIND OF SPA EXPERIENCE WHAT TO EXPECT
 SOLUN HOTEL & SPA **** SUPERIOR Ul.Nikola Vapcarov br.10 1000 Skopje Macedonia tel:+389 2 32 32 512 Mob:+389 78 999992 Fax:+389 2 32 32 513 info@hotelsolun.com.mk bookingonline@hotelsolun.com.mk www.hotelsolun.com
SOLUN HOTEL & SPA **** SUPERIOR Ul.Nikola Vapcarov br.10 1000 Skopje Macedonia tel:+389 2 32 32 512 Mob:+389 78 999992 Fax:+389 2 32 32 513 info@hotelsolun.com.mk bookingonline@hotelsolun.com.mk www.hotelsolun.com
VALIDITY FROM : TO MAINTENANCE SERVICES 2018
 VALIDITY FROM : 01-01-2018 TO 31-12-2018 FOR MAINTENANCE STATIONS: STARTING SUMMER 18: DUS DUSSELDORF AIRPORT TXL BERLIN TEGEL AIRPORT MUC MUNICH AIRPORT CGN COLOGNE AIRPORT LEJ LEIPZIG AIRPORT BRE BREMEN
VALIDITY FROM : 01-01-2018 TO 31-12-2018 FOR MAINTENANCE STATIONS: STARTING SUMMER 18: DUS DUSSELDORF AIRPORT TXL BERLIN TEGEL AIRPORT MUC MUNICH AIRPORT CGN COLOGNE AIRPORT LEJ LEIPZIG AIRPORT BRE BREMEN
HOSE ASSEMBLY CLEANLINESS
 November 18, 2002 HOSE ASSEMBLY CLEANLINESS Preface Hydraulic system cleanliness is a term used to describe the level of solid and liquid contamination found in hydraulic systems. Contamination may be
November 18, 2002 HOSE ASSEMBLY CLEANLINESS Preface Hydraulic system cleanliness is a term used to describe the level of solid and liquid contamination found in hydraulic systems. Contamination may be
Critical environment products
 Critical environment products Where clean is critical every container, every time The only thing in your container is what you add Clean process products for critical environments Working within a critical
Critical environment products Where clean is critical every container, every time The only thing in your container is what you add Clean process products for critical environments Working within a critical
THE NEXT GENERATION OF WATER COOLERS. VonTaine HRC - The complete range. ProAcqua Group. a ProMinent Company
 THE NEXT GENERATION OF WATER COOLERS VonTaine HRC - The complete range ProAcqua Group a ProMinent Company Printed in Italy, The ultimate in water coolers Well insulate ice-bank for high volume supply of
THE NEXT GENERATION OF WATER COOLERS VonTaine HRC - The complete range ProAcqua Group a ProMinent Company Printed in Italy, The ultimate in water coolers Well insulate ice-bank for high volume supply of
pulverisette 19 crushing sample preparation cutting mill for your lab Universal Cutting Mill
 pulverisette 19 Universal Cutting Mill Suitable for grinding soft to medium-hard, dry materials, including fibrous materials and plastics Very simple to use and easy to clean Computer-optimised cutting
pulverisette 19 Universal Cutting Mill Suitable for grinding soft to medium-hard, dry materials, including fibrous materials and plastics Very simple to use and easy to clean Computer-optimised cutting
TOTAL COLIFORM ANDE.coli INDICATOR BACTERIA TEST KIT UV
 TOTAL COLIFORM ANDE.coli INDICATOR BACTERIA TEST KIT 4-3616-UV blank WARNING! This set contains chemicals that may be harmful if misused. Read cautions on individual containers carefully. Not to be used
TOTAL COLIFORM ANDE.coli INDICATOR BACTERIA TEST KIT 4-3616-UV blank WARNING! This set contains chemicals that may be harmful if misused. Read cautions on individual containers carefully. Not to be used
PRODUCT NO.:
 PRODUCT NO.: 7182-01 STARTER KIT (Germfree) For GermFree Glove Box DESCRIPTION: This convenient starter kit will allow the operator to jumpstart their cleaning with the GermFree Compounding Aseptic Isolator
PRODUCT NO.: 7182-01 STARTER KIT (Germfree) For GermFree Glove Box DESCRIPTION: This convenient starter kit will allow the operator to jumpstart their cleaning with the GermFree Compounding Aseptic Isolator
EB300. Ethanol Burner. User s Manual Installation Instructions
 EB300 Ethanol Burner User s Manual Installation Instructions EB300 Burner Burner Lid User s Manual What s in the box: 1pc 1pc 1pc Installation Instructions EB300 - User s Manual The Ignis Ethanol Burner
EB300 Ethanol Burner User s Manual Installation Instructions EB300 Burner Burner Lid User s Manual What s in the box: 1pc 1pc 1pc Installation Instructions EB300 - User s Manual The Ignis Ethanol Burner
J.M. Marques a, C. Matos b, P.M. Carreira c and M.O. Neves a
 GEOCHEMICAL AND ISOTOPIC TOOLS TO ASSESS CALDAS DA RAINHA THERMOMINERAL WATER SYSTEM ASCRIBED TO A KARST/FISSURED-POROUS ENVIRONMENT (PORTUGAL): A REVIEW J.M. Marques a, C. Matos b, P.M. Carreira c and
GEOCHEMICAL AND ISOTOPIC TOOLS TO ASSESS CALDAS DA RAINHA THERMOMINERAL WATER SYSTEM ASCRIBED TO A KARST/FISSURED-POROUS ENVIRONMENT (PORTUGAL): A REVIEW J.M. Marques a, C. Matos b, P.M. Carreira c and
FORM (Apr. 2006)
 2212 Slcier MODEL 2212 SLICER MODEL 2212 ML-136132 701 S. RIDGE AVENUE TROY, OHIO 45374-0001 937 332-3000 www.hobartcorp.com FORM 35215 (Apr. 2006) Installation, Operation, and Care of MODEL 2212 Slicer
2212 Slcier MODEL 2212 SLICER MODEL 2212 ML-136132 701 S. RIDGE AVENUE TROY, OHIO 45374-0001 937 332-3000 www.hobartcorp.com FORM 35215 (Apr. 2006) Installation, Operation, and Care of MODEL 2212 Slicer
Plastic Ware CODE DESCRIPTION QTY
 Plastic Ware CODE DESCRIPTION QTY 1 BK044-2 GRADUATED PLASTIC BEAKERS, 50ML 1 2 BK044-3 GRADUATED PLASTIC BEAKERS, 100ML 1 3 BK044-4 GRADUATED PLASTIC BEAKERS, 250ML 1 4 BK044-5 GRADUATED PLASTIC BEAKERS,
Plastic Ware CODE DESCRIPTION QTY 1 BK044-2 GRADUATED PLASTIC BEAKERS, 50ML 1 2 BK044-3 GRADUATED PLASTIC BEAKERS, 100ML 1 3 BK044-4 GRADUATED PLASTIC BEAKERS, 250ML 1 4 BK044-5 GRADUATED PLASTIC BEAKERS,
Emergency Disinfection of Drinking Water
 Emergency Disinfection of Drinking Water USE ONLY WATER THAT HAS BEEN PROPERLY DISINFECTED FOR DRINKING, COOKING, MAKING ANY PREPARED DRINK, OR FOR BRUSHING TEETH Use bottled water that has not been exposed
Emergency Disinfection of Drinking Water USE ONLY WATER THAT HAS BEEN PROPERLY DISINFECTED FOR DRINKING, COOKING, MAKING ANY PREPARED DRINK, OR FOR BRUSHING TEETH Use bottled water that has not been exposed
Supplementary Figure 1. HR-TEM images of unzipped carbon nanostructures by N- dopant-specific unzipping of NCNTs. a, Sequential unzipping of inner
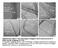 Supplementary Figure 1. HR-TEM images of unzipped carbon nanostructures by N- dopant-specific unzipping of NCNTs. a, Sequential unzipping of inner wall of NCNTs b,c, Unzipped nanostructures consisting
Supplementary Figure 1. HR-TEM images of unzipped carbon nanostructures by N- dopant-specific unzipping of NCNTs. a, Sequential unzipping of inner wall of NCNTs b,c, Unzipped nanostructures consisting
Herolily USA Inc., headquartered in Los Angeles, California was the first company to introduce cuttingedge, game-changing cold packs to the USA
 1 Herolily USA Inc., headquartered in Los Angeles, California was the first company to introduce cuttingedge, game-changing cold packs to the USA market. Herolily ice products provide vast technology improvements
1 Herolily USA Inc., headquartered in Los Angeles, California was the first company to introduce cuttingedge, game-changing cold packs to the USA market. Herolily ice products provide vast technology improvements
Instruction Manual please read before use
 Instruction Manual please read before use XL Burner (patent pending) 307 Tchoupitoulas Street, Suite 100 New Orleans, LA 70130 p : 888.596.6960 504.596.6960 e : office@brasafire.com www.brasa.co Item Identification
Instruction Manual please read before use XL Burner (patent pending) 307 Tchoupitoulas Street, Suite 100 New Orleans, LA 70130 p : 888.596.6960 504.596.6960 e : office@brasafire.com www.brasa.co Item Identification
Structures of Solids. Prof Andrew Goodwin Michaelmas 2014
 Structures of Solids Prof Andrew Goodwin Michaelmas 2014 Li + Be 2+ N 3 O 2 F r/n ~ Polarisability Li + B C 76 22 48 70 133 Na + Mg 2+ Al 3+ Si 4+ P 3 S 2 Cl 102 36 18 10 70 92 181 K + Ca 2+ Sc 3+ Ti 3+
Structures of Solids Prof Andrew Goodwin Michaelmas 2014 Li + Be 2+ N 3 O 2 F r/n ~ Polarisability Li + B C 76 22 48 70 133 Na + Mg 2+ Al 3+ Si 4+ P 3 S 2 Cl 102 36 18 10 70 92 181 K + Ca 2+ Sc 3+ Ti 3+
Macdonald et al., Fig_DR1.pdf
 DR2010028 Figure DR1 - Location map of the Tatonduk Inlier with inset of geological map of the Tatonduk Inlier modified from Brabb & Churkin (1969) and Van Kooten et al. (1997); NAM-ancestral North America,
DR2010028 Figure DR1 - Location map of the Tatonduk Inlier with inset of geological map of the Tatonduk Inlier modified from Brabb & Churkin (1969) and Van Kooten et al. (1997); NAM-ancestral North America,
Innovative Products from the
 Innovative Products from the #1 Ranked Nonstick Cookware Real diamond particles in a high-quality nonstick formula. Cast aluminum heats evenly and will never warp. Made in Switzerland Ideal for healthy
Innovative Products from the #1 Ranked Nonstick Cookware Real diamond particles in a high-quality nonstick formula. Cast aluminum heats evenly and will never warp. Made in Switzerland Ideal for healthy
Experimental Procedure. Lab 402
 Experimental Procedure Lab 402 A. Sample Preparation 1. Prepare a clean crucible Support the crucible and lid on a clay triangle and heat with an intense flame for 5 minutes. Clay triangle Allow them to
Experimental Procedure Lab 402 A. Sample Preparation 1. Prepare a clean crucible Support the crucible and lid on a clay triangle and heat with an intense flame for 5 minutes. Clay triangle Allow them to
Type Acceptance Report
 TAR 9/21B/12 CESSNA 120/140 Aircraft Certification Unit TABLE OF CONTENTS EXECUTIVE SUMMARY 1 1. INTRODUCTION 1 2. ICAO TYPE CERTIFICATE DETAILS 1 3. TYPE ACCEPTANCE DETAILS 2 4. NZCAR 21.43 DATA REQUIREMENTS
TAR 9/21B/12 CESSNA 120/140 Aircraft Certification Unit TABLE OF CONTENTS EXECUTIVE SUMMARY 1 1. INTRODUCTION 1 2. ICAO TYPE CERTIFICATE DETAILS 1 3. TYPE ACCEPTANCE DETAILS 2 4. NZCAR 21.43 DATA REQUIREMENTS
STUDY OF ALAQI SECONDARY CHANNEL IN LAKE NASSER
 ABSTRACT STUDY OF ALAQI SECONDARY CHANNEL IN LAKE NASSER GamalSallam 1, Mohamed Ihab 2, and Waleed Emary 3 Associate Professor, National Water Research Center, Email: gasallam@yahoo.com Civil Engineer,
ABSTRACT STUDY OF ALAQI SECONDARY CHANNEL IN LAKE NASSER GamalSallam 1, Mohamed Ihab 2, and Waleed Emary 3 Associate Professor, National Water Research Center, Email: gasallam@yahoo.com Civil Engineer,
Realax hoses & tubes specification. Fitted for the purpose
 Hoses pumps and tubes for APY, Series ISI, RP IP high and pressure RP series Realax hoses & tubes specification Hose pumps, also called peristaltic pumps, are excellent for moving a wide variety of fluids
Hoses pumps and tubes for APY, Series ISI, RP IP high and pressure RP series Realax hoses & tubes specification Hose pumps, also called peristaltic pumps, are excellent for moving a wide variety of fluids
I. PREPARING FOR TESTING
 Guidelines for Testing Charcoal Stoves with WBT 4.2.2 June 14, 2013 The following guidelines are intended to improve repeatability and reduce variability both from test to test and from site to site. The
Guidelines for Testing Charcoal Stoves with WBT 4.2.2 June 14, 2013 The following guidelines are intended to improve repeatability and reduce variability both from test to test and from site to site. The
There are 7 kinds of unique dry medium for hygienic testing and detection of food poisoning bacteria.
 Simple and Easy Dry Media for Microbial Count and Detection There are 7 kinds of unique dry medium for hygienic testing and detection of food poisoning bacteria. s Small and compact dry media (sterilized)
Simple and Easy Dry Media for Microbial Count and Detection There are 7 kinds of unique dry medium for hygienic testing and detection of food poisoning bacteria. s Small and compact dry media (sterilized)
Österreichisches Technolgiesymposium Oktober 2016, TU Wien. Ion Mobility - from Research to Routine
 Österreichisches Technolgiesymposium 2016 25. Oktober 2016, TU Wien Ion Mobility - from Research to Routine Dr. Mathias Hofmann European Applications Field Support Manager 2016 Waters Corporation 1 2016
Österreichisches Technolgiesymposium 2016 25. Oktober 2016, TU Wien Ion Mobility - from Research to Routine Dr. Mathias Hofmann European Applications Field Support Manager 2016 Waters Corporation 1 2016
